Recommendations
Key Recommendations
Early recognition and rapid diagnosis are essential to prevent transmission and provide supportive care in a timely manner. Have a high index of clinical suspicion for COVID-19 in all patients who present with fever and/or acute respiratory illness; however, be aware that some patients may not present with signs or symptoms of a febrile respiratory illness.
COVID-19 is a notifiable disease. Report all suspected or confirmed cases to your local health authorities. Isolate all suspected or confirmed cases immediately and follow local infection prevention and control guidelines.
Suspect the diagnosis in patients with the following signs/symptoms: a new continuous cough, fever, altered sense of taste or smell, sore throat, fatigue, headache, dyspnoea, myalgia, arthralgia, rhinorrhoea, nasal congestion, sneezing, malaise, expectoration, or chest tightness/pain, particularly if the person has been in contact with a suspected or confirmed case. Patients may also present with gastrointestinal, cutaneous, or ocular symptoms.[435][436]
Be on high alert for children and adolescents with acute gastrointestinal symptoms and signs of cardiac inflammation. Evidence so far suggests a milder or asymptomatic course of disease in children and adolescents.[437] However, a rare multisystem inflammatory condition has been reported.[438]
Order a real-time reverse transcription polymerase chain reaction (RT-PCR) to confirm the diagnosis. Upper and lower respiratory specimens are preferred.[439] Rapid antigen tests may be recommended in certain circumstances.[440][441][442]
Order the following laboratory investigations in hospitalised patients: full blood count, comprehensive metabolic panel, arterial blood gas, thyroid function tests, blood glucose level, coagulation screen, inflammatory markers, cardiac biomarkers, serum creatine kinase, and blood and sputum cultures for other pathogens. Pulse oximetry may reveal low oxygen saturation.
Prioritise a chest x-ray in patients who are seriously ill with suspected pneumonia. Consider a computed tomography (CT) scan of the chest if chest x-ray is uncertain or normal.[443] Consult local guidelines as imaging recommendations may vary.
COVID-19 care pathways should be established at local, regional, and national levels for people with suspected or confirmed COVID-19.
Screen patients at the first point of contact within the health system based on case definitions and an assessment of symptoms, and enter suspected or confirmed cases into the pathway.[444]
Immediately isolate all suspected and confirmed cases and implement local infection prevention and control procedures.
Take a detailed history to ascertain the level of risk for COVID-19 and assess the possibility of other causes, including a travel history and an assessment of risk factors.
Suspect the diagnosis in:[124]
People residing or working in an area with a high risk of transmission (e.g., closed settings, humanitarian settings) and people working in a health setting (including within health facilities and households) at any time within the 14 days prior to symptom onset
People who have had contact with a probable or confirmed case. A contact is a person who has experienced any one of the following exposures:
Face-to-face contact with a probable or confirmed case within 1 metre (3 feet) and for at least 15 minutes
Direct physical contact with a probable or confirmed case
Direct care for a patient with probable or confirmed COVID-19 without using recommended personal protective equipment
Other situations as determined by local health authorities based on local risk assessments
Exposure must have occurred during the infectious period of the case. For symptomatic cases, this means 2 days before and 10 days after symptom onset of the case, plus at least 3 additional days without symptoms, for a minimum of 13 days total after symptom onset. For asymptomatic cases, this means 2 days before and 10 days after the date on which the sample that led to confirmation was taken.[124]
Some patients may be minimally symptomatic or asymptomatic, while others may present with severe pneumonia or complications such as acute respiratory syndrome, septic shock, acute myocardial infarction, venous thromboembolism, or multi-organ failure.
The majority of patients have mild illness that does not warrant medical intervention or hospitalisation, depending on the circulating SARS-CoV-2 variant.
The pooled proportion of non-severe illness in people infected with the Omicron variant was 98%, and the pooled proportion of asymptomatic infection was 25% (proportions varied depending on vaccination status).[445]
Common symptoms include:
Fever
Cough
Fatigue
Myalgia
Arthralgia
Dyspnoea
Altered sense of taste/smell
Sore throat
Headache
Rhinorrhoea
Nasal congestion
Sneezing
Expectoration
Less common or uncommon symptoms include:
Chest tightness/pain
Malaise
Dizziness
Confusion
Delirium
Gastrointestinal symptoms
Cutaneous symptoms
Ocular symptoms
Haemoptysis
Audio-vestibular symptoms
Oral mucosal lesions
For more detailed information on signs and symptoms, see History and exam.
No single sign or symptom can accurately diagnose COVID-19, and neither the absence or presence of specific signs or symptoms are accurate enough to rule in or rule out disease.
A Cochrane review found that the presence of anosmia and/or ageusia may be useful as a red flag for diagnosis. Cough or fever may also increase the probability of diagnosis to an extent that is clinically relevant and should prompt further testing. Other symptoms increase the probability of the diagnosis to a much lesser extent.[446]
Signs and symptoms may differ in the context of circulating SARS-CoV-2 variants.
Symptoms that characterise infection with the Omicron variant differ moderately from those that characterised infection with the Delta variant. There has been a shift towards predominantly upper respiratory tract symptoms with the Omicron variant and its subvariants.[447]
The most common symptoms of Omicron infection according to data from the UK COVID Symptom Study were rhinorrhoea, headache, fatigue, sneezing, and sore throat.[448]
Sore throat and hoarse voice were consistently more prevalent among people with Omicron infection compared with those with Delta infection. Loss of/altered smell, eye soreness/burning, sneezing, headache, fever, dizziness, and brain fog were significantly less prevalent among people with Omicron infection compared with those with Delta infection. Loss of smell, a pathognomonic feature of previous SARS-CoV-2 variants, was present in only <20% of cases of people infected with the Omicron variant.[449]
In the later stages of the pandemic, the sensitivity of fever and cough in predicting infection decreased, and the sensitivity of other symptoms (e.g., sore throat, sputum production, headache, myalgia, fatigue, dyspnoea, diarrhoea) increased.[450]
Pregnant women generally present in a similar way to non-pregnant people.
The clinical characteristics in pregnant women are similar to those reported for non-pregnant adults.[451]
The most common symptoms in pregnant women are fever and cough. However, pregnant women are less likely to report fever, dyspnoea, and myalgia compared with non-pregnant women of reproductive age. Pregnant and recently pregnant women were more likely to be asymptomatic than non-pregnant women of reproductive age.[218][219]
It is important to note that symptoms such as fever, dyspnoea, gastrointestinal symptoms, and fatigue may overlap with symptoms due to physiological adaptations of pregnancy or adverse pregnancy events.
Atypical presentations have been reported.
Atypical presentations may occur, especially in older patients and patients who are immunocompromised (e.g., fatigue, reduced alertness, diarrhoea, loss of appetite, delirium, reduced mobility, absence of fever).[444]
There have been case reports of parotitis (possibly related to intraparotid lymphadenitis), oral vesiculobullous lesions, persistent hiccups, and androgenetic alopecia in patients with COVID-19; however, it is unknown whether there is a causal association.[452][453][454][455]
Co-infections are possible.
Co-infections can be:
Co-infections at presentation
Reactivation of latent infections
Nosocomial infections
Opportunistic fungal infections
The pooled prevalence of co-infection in SARS-CoV-2-positive patients was 19%, with viral coinfections being more common than bacterial and fungal co-infections.[456]
The most frequently identified bacteria were Klebsiella pneumonia, Streptococcus pneumoniae, and Staphylococcus aureus.
The most frequently identified viruses were influenza type A, influenza type B, and respiratory syncytial virus. Co-infection with influenza may increase the risk for intensive care unit admission and mechanical ventilation, but does not increase the risk of mortality.[457]
The most frequently identified fungi was Aspergillus.
Co-infection with tuberculosis, HIV, and malaria have been reported.[458][459][460]
Signs and symptoms may be similar to other common viral infections and other childhood illnesses (e.g., influenza, respiratory syncytial virus infection, gastroenteritis), so a high index of suspicion for COVID-19 is required in children and adolescents.[461]
Children and adolescents usually have fewer and milder symptoms, and they are less likely to progress to severe disease compared with adults.[20][462] Children and adolescents are also more likely to be asymptomatic.[463] The reasons for this are still under investigation, but children appear to develop a higher and more sustained antibody response compared with adults.[464]
The frequency of severe disease in children was lower with infection with the Omicron variant compared with Delta and Alpha variants.[465]
Early studies suggested a higher risk of severe or critical disease in infants <1 year of age compared with children of other age groups; however, the studies had limitations and there is no conclusive evidence that younger age is a risk factor for severe disease in children and adolescents.[20]
The most common symptoms in neonates include fever, inability to feed, lethargy, irritability, feeding difficulties, dyspnoea, silent hypoxia, and neurological symptoms. Cases of late-onset neonatal sepsis and encephalitis have been reported rarely.[437][466][467][468]
Be alert for signs and symptoms of multisystem inflammatory syndrome in children (MIS-C), also known as paediatric inflammatory multisystem syndrome (PIMS).
Consider MIS-C in children presenting with fever and abdominal symptoms, particularly if they develop conjunctivitis or a rash. Refer to a paediatric accident and emergency department for evaluation.[469] For more information, see Complications.
Co-infections are possible in children.
Co-infections have been documented in 9% of children, with the most common pathogens being respiratory syncytial virus and Mycoplasma pneumoniae. Co-infections are less common in children compared with adults.[470]
Viral co-infection has been associated with a higher frequency of severe outcomes in children, particularly in infants.[465] A higher percentage of children with influenza and SARS-CoV-2 coinfection required non-invasive or invasive respiratory support compared with those with influenza only.[471]
Perform a physical examination.
Patients may be febrile (with or without chills/rigors) and have obvious cough and/or difficulty breathing.
Auscultation of the chest may reveal inspiratory crackles, rales, and/or bronchial breathing in patients with pneumonia or respiratory distress. Use caution when auscultating patients given the risk for cross-contamination. Clean the stethoscope properly between uses.[472]
Patients with respiratory distress may have tachycardia, tachypnoea, or cyanosis accompanying hypoxia. Bradycardia has been noted in a small cohort of patients with mild to moderate disease.[473]
Pulse oximetry may reveal low oxygen saturation.
Hypoxia (hypoxaemia) is defined as oxygen saturation <94%, or <88% in the presence of chronic lung disease.[474] However, the oxygen saturation cut-offs used to define disease severity in patients with COVID-19 vary and you should consult your local guidelines.
The World Health Organization (WHO) defines severe disease as SpO2 <90%.[444]
In the UK, the National Institute for Health and Care Excellence (NICE) recommends using current NHS guidelines.[405] A reading of <92% is one defining feature of severe disease that requires urgent hospital admission, while a reading of 93% to 94% may indicate moderate disease.[475]
In the US, the Infectious Diseases Society of America (IDSA) defines severe disease as SpO2 <94%.[401]
Clinicians should be aware that patients with COVID-19 can develop ‘silent hypoxia': their oxygen saturations can drop to low levels and precipitate acute respiratory failure without the presence of obvious symptoms of respiratory distress.[476]
Pulse oximeters may exhibit suboptimal accuracy in certain populations, especially in those who have darker skin. Always interpret SpO2 within the context of a patient’s clinical presentation.
A systematic review and meta-analysis found that pulse oximetry probably overestimates oxygen saturation compared with SaO2 (arterial oxygen saturation) in people with high levels of skin pigmentation and people who report their ethnicity as black/African-American (moderate- and low-certainty evidence). The bias of measurements for people with other levels of skin pigmentation or those from other ethnic groups is either more uncertain or suggests no overestimation.[477]
The US Food and Drug Administration (FDA) has warned that multiple factors can affect the accuracy of a pulse oximeter reading (e.g., poor circulation, skin pigmentation, skin thickness, skin temperature, current tobacco use, use of fingernail polish). The FDA recommends considering accuracy limitations when using a pulse oximeter to assist in diagnosis and treatment decisions, and to use trends in readings over time rather than absolute cut-offs if possible.[478]
Pulse oximeters can be used at home to detect hypoxia in patients with mild to moderate disease.
Evidence suggests that patients who may benefit most from monitoring are those who are symptomatic and are either over age 65 years, or are under age 65 years and are extremely clinically vulnerable to COVID-19.[475]
Home pulse oximetry requires clinical support (e.g., regular phone contact from a health professional in a virtual ward setting).
Traditional methods of recognising further deterioration may not help predict those patients who go on to develop respiratory failure.
While National Early Warning Score 2 (NEWS2) is still recommended for use in patients with COVID-19, the UK Royal College of Physicians now advises that any increase in oxygen requirements in these patients should trigger an escalation call to a competent clinical decision maker, and prompt an initial increase in observations to at least hourly until a clinical review happens.[479]
Only a small proportion of patients have other organ dysfunction, meaning that after the initial phase of acute deterioration, traditional methods of recognising further deterioration (e.g., NEWS2 scores) may not help predict those patients who go on to develop respiratory failure.[476]
A systematic review and meta-analysis found that the NEWS2 score had moderate sensitivity and specificity in predicting the deterioration of patients with COVID-19. The score showed good discrimination in predicting the combined outcome of the need for intensive respiratory support, admission to the intensive care unit, or in-hospital mortality.[480]
A NEWS2 score ≥4 was found to be the best cut-off point for predicting respiratory failure.[481] However, you should consult your local guidance.
For more information on medical early warning and prognostic scores, see Monitoring.
Order the following laboratory investigations in all patients with severe disease:
ABG
FBC
Comprehensive metabolic panel
Blood glucose level
Coagulation screen
Inflammatory markers (e.g., serum C-reactive protein [CRP], erythrocyte sedimentation rate, lactate dehydrogenase, procalcitonin, and ferritin)
Cardiac biomarkers
The most common laboratory abnormalities are:[482][483][484]
Lymphopenia
Leukocytosis
Leukopenia
Thrombocytopenia
Hypoalbuminaemia
Elevated cardiac biomarkers
Elevated inflammatory markers
Elevated D-dimer
Abnormal liver and renal function
For more detailed information on tests and test results, see Investigations.
Laboratory abnormalities – in particular, lymphopenia, leukocyte abnormalities, and other markers of systemic inflammation – are less common in children.[485][486] Most patients (62%) with asymptomatic disease present with normal laboratory parameters. Of those with laboratory abnormalities, leukopenia, lymphopenia, elevated lactate dehydrogenase, and elevated C-reactive protein were the most common findings.[487]
A Cochrane review found that while routine laboratory tests used to assess blood clotting and inflammation (i.e., CRP, lactate dehydrogenase, D-dimer, lymphocyte count, neutrophil to lymphocyte ratio) may be used for initial risk stratification (along with clinical signs and symptoms) to assess a patient, none of these tests performed well enough to safely rule out progression to severe or critical disease.[488]
Collect blood and sputum specimens for culture in patients with severe or critical disease.
Cultures are required to rule out other causes of lower respiratory tract infection and sepsis. Specimens should be collected prior to starting empirical antimicrobials if possible.[444]
Patients may develop bacterial or fungal co-infections; therefore, it is important to ensure appropriate microbiological specimens are taken and imaging is ordered, according to local guidelines, when co-infections are suspected.
How to obtain an arterial blood sample from the radial artery.
There are three main methods for detecting SARS-CoV-2 infection:
Molecular testing
Rapid antigen testing
Serological testing
The choice of which test to use in which setting requires careful consideration of the purpose of testing and the resources available, while also balancing test characteristics of accuracy, accessibility, affordability, and the rapidity with which results are needed.[489]
Molecular tests: highly specific and sensitive at detecting viral RNA, and are the preferred test for confirming diagnosis in symptomatic people. However, these tests are expensive and require specialised skills and instruments, and results can take up to 24 to 48 hours.
Rapid antigen tests: detect viral protein and are less sensitive than molecular tests, but are faster, easier, and cheaper, and are able to detect infection in those who are most likely to be at risk of transmitting the virus.
Serological tests: may be used to establish a late or retrospective diagnosis if molecular and rapid antigen tests are both negative, or may be useful surveillance tools to inform public policy.
A combination of tests may be required depending on individual patient circumstances (e.g., a symptomatic person with a negative rapid antigen test may require confirmation testing with a molecular test).
The diagnostic accuracy of molecular tests and rapid antigen tests does not appear to be influenced by the Omicron variant.[490]
A prospective cohort study found that the performance of rapid antigen tests in people infected with the Omicron variant is not inferior to that in people infected with the Delta variant.[491]
Testing strategies vary widely between countries, and you should consult your local public health authority for advice when deciding which test to use.
The World Health Organization (WHO) recommends the following principles as part of testing programmes:[492]
Test early in the course of COVID-19 to enable timely care and treatment.
Test to reduce spread.
Test to track the evolution of the epidemic and the SARS-CoV-2 virus.
Widespread screening of asymptomatic people is no longer recommended in most countries. For more information, see Screening.
The World Health Organization (WHO) recommends testing all people who meet the suspected case definition, regardless of vaccination status or disease history.[493]
When resources are constrained, suspected cases who are at risk of developing severe disease, those who require hospitalisation, and the first symptomatic individuals in the setting of a suspected outbreak should be prioritised.
Testing of asymptomatic individuals is currently recommended only for specific people with a risk of exposure including contacts of confirmed or probable cases and frequently exposed occupational groups such as healthcare workers and long-term care facility workers.
In the UK, from 1 April 2024, testing is recommended for the following situations in NHS settings:[494]
Symptomatic adults and children admitted for care or developing symptoms within hospitals where having COVID-19 will change clinical management: for example, to inform treatment (PCR and lateral flow device at local discretion)
Symptomatic adults and children eligible for antiviral treatment or in community settings to inform treatment (lateral flow device)
Discharge of asymptomatic patients to care settings such as care homes or hospices (not routinely required; however, lateral flow devices could be introduced at local discretion based on risk)
Symptomatic NHS staff and staff in NHS-commissioned independent healthcare providers working on wards focused on treating severely immunosuppressed individuals (lateral flow device in line with locally-derived protocols)
Outbreak testing in healthcare settings (lateral flow device or PCR in line with locally-directed protocols; PCR where needed for diagnosis)
Testing recommendations for care services (adult social care and hospices) and other non-healthcare settings are also available.
In the US, testing may be considered in any person with signs or symptoms consistent with COVID-19.[442]
The choice of test depends on the clinical scenario. Consult your local public health authority for guidance as testing priorities depend on local recommendations and available resources, and guidance may update regularly.
Molecular testing is recommended to confirm the diagnosis.
Order a nucleic acid amplification test, such as RT-PCR, for SARS-CoV-2 in patients with suspected infection, whenever possible.[439]
Tests should be performed according to guidance issued by local health authorities and adhere to appropriate biosafety practices.
Commonly used assays are expected to be able to detect SARS-CoV-2 variants. However, some tests may be impacted by variants.[495]
Specimens
Collect specimens under appropriate infection prevention and control procedures.
The optimal specimen for testing depends on the clinical presentation and the time since symptom onset. The World Health Organization (WHO) recommends the following.[439]
Upper respiratory specimens: recommended for early-stage infections, especially asymptomatic or mild cases. Nasopharyngeal swabs yield a more reliable result than oropharyngeal swabs; combined nasopharyngeal and oropharyngeal swabs further improve reliability.
Lower respiratory specimens: recommended for later-stage infections, or patients in whom there is a strong suspicion for infection and their upper respiratory tract specimen test was negative. Suitable specimens are sputum and/or endotracheal aspirate or bronchoalveolar lavage in patients with more severe respiratory disease. However, consider the high risk of aerosol transmission when collecting lower respiratory specimens – an induced sputum specimen is not recommended as it may increase the risk of aerosol transmission.
Other respiratory specimens: studies on combined oropharyngeal and nares/nasal swabs, mid-turbinate or lower nasal or nares swabs, or tongue swabs have been conducted; however, further assessment and validation is required. Oral fluid collection may be suitable in some circumstances (e.g., young children, older patients with dementia).
Saliva specimens: not currently recommended as the sole specimen type for routine clinical diagnostics.
Faecal specimens: consider when upper or lower respiratory specimens are negative and the clinical suspicion for infection remains (may be used from the second week after symptom onset).
Recommended specimen types may differ between countries. For example, in the US, the Centers for Disease Control and Prevention (CDC) recommends the following specimens:[496]
Upper respiratory specimens: nasopharyngeal or oropharyngeal swab; nasal mid-turbinate swab; anterior nares swab; or nasopharyngeal/nasal wash/aspirate.
Lower respiratory specimens: sputum, bronchoalveolar lavage, tracheal aspirate, pleural fluid, or lung biopsy.
Complications associated with nasal swab testing are not well characterised and data is scarce.
Complications were extremely low in one study (1.24 complications per 100,000 tests). Adverse effects may include epistaxis, nasal discomfort, headache, ear discomfort, rhinorrhoea, and broken swabs being stuck (and requiring removal via nasal endoscopy). Bleeding may be life-threatening. Correct sampling techniques are crucial.[497][498]
Cases of iatrogenic cerebrospinal fluid leak have been reported after nasal testing in people with undiagnosed skull base defects and people with no pre-existing skull base conditions.[499][500] The majority of cases (59%) had no reported predisposing factor in their history. Surgical repair is the recommended treatment option.[501]
Test result
A positive RT-PCR result confirms SARS-CoV-2 infection (in the context of the limitations associated with RT-PCR testing). If the result is negative, and there is still a clinical suspicion of infection (e.g., an epidemiological link, typical x-ray findings, absence of another aetiology), resample the patient and repeat the test. A positive result confirms infection. If the second test is negative, consider serological testing (see below).[439] The pooled sensitivity has been estimated to be 87.8%, with the specificity estimated to be in the range of 87.7% to 100%.[502]
Genomic sequencing is not routinely recommended, but may be useful to investigate the dynamics of an outbreak, including changes in the size of an epidemic over time, its spatiotemporal spread, and testing hypotheses about transmission routes.[439] It is also useful in the context of circulating SARS-CoV-2 variants to differentiate between variants.
See the Testing limitations and evidence section (below).
Testing for other infections
Collect nasopharyngeal swabs for testing to rule out infection with other respiratory pathogens (e.g., influenza, atypical pathogens) when clinically indicated according to local guidance. Depending on local epidemiology and clinical symptoms, test for other potential causes including malaria, dengue fever, and tuberculosis as appropriate.[444] It is important to note that co-infections can occur, and a positive test for a non-COVID-19 pathogen does not rule out COVID-19.
Antigen testing relies on direct detection of SARS-CoV-2 viral proteins in upper respiratory specimens or saliva using a lateral flow immunoassay.[440]
While antigen tests are substantially less sensitive than RT-PCR, particularly in asymptomatic people, they offer the possibility of rapid, inexpensive, and early detection of the most infectious cases in appropriate settings.
Antigen testing is recommended in settings likely to have the most impact on early detection of cases for care and contact tracing, and where test results are most likely to be correct.
Results are usually available in less than 30 minutes.
Rapid, lateral flow antigen tests for home use are available over-the-counter in some countries. Laboratory-based (non-rapid) antigen tests may also be available in some countries.
International guidelines for the use of rapid antigen tests vary. Consult your local guidance.
The World Health Organization (WHO) recommends antigen testing for primary case detection, for contact tracing, during outbreak investigations, and to monitor trends of disease incidence in communities.[440][503]
Tests should meet the minimum performance requirements of ≥80% sensitivity and ≥97% specificity compared with an RT-PCR reference assay.
Antigen testing should be prioritised for use in symptomatic people who meet the case definition in the first 5 to 7 days of symptom onset, and to test asymptomatic people at high risk of infection, including contacts and health workers, particularly in settings where molecular testing capacity is limited.
Results are most reliable in areas where there is ongoing community transmission.
Self-testing should be offered in addition to professionally administered testing services. It should always be voluntary and never mandatory or coercive.
In the US, the Centers for Disease Control and Prevention (CDC) recommends that antigen tests may be used for certain testing situations; however, confirmatory molecular testing may be needed.[442]
See the Testing limitations and evidence section (below).
Serology cannot be used as a standalone diagnostic test for acute SARS-CoV-2 infections, and should not be used to establish the presence or absence of acute infection.
However, it may be useful in various settings (e.g, negative molecular testing, diagnosing patients with late presentation or prolonged symptoms, serosurveillance studies).[439][504]
Antibody responses to SARS-CoV-2 typically occur during the first 1 to 3 weeks of illness, with the seroconversion time of IgG antibodies often being earlier than that of IgM antibodies.[505][506]
Results are usually available in up to 24 hours.
International guidelines for the use of serological tests vary. Consult your local guidance.
The World Health Organization (WHO) recommends collecting a paired serum sample, one specimen in the acute phase and one in the convalescent phase 2 to 4 weeks later, in patients where infection is strongly suspected and the RT-PCR result is negative.[439]
Seroconversion or a rise in antibody titres in paired sera help to confirm whether the infection is recent and/or acute. If the initial sample tests positive, this could be due to a past infection that is not related to the current illness.
Seroconversion may be faster and more robust in patients with severe disease compared with those with mild disease or asymptomatic infection.
The Infectious Diseases Society of America (IDSA) recommends against using serological testing to diagnose infection during the first 2 weeks following symptom onset or to provide evidence of infection in symptomatic patients with a high clinical suspicion and repeatedly negative molecular tests. Serological testing may be used to assist with the diagnosis of multisystem inflammatory syndrome in children or when evidence of previous infection is desired.[507]
BMJ practice pointer: testing for SARS-CoV-2 antibodies Opens in new window
Rapid antibody detection tests
While rapid antibody detection kits have been approved for the qualitative detection of SARS-CoV-2 IgG/IgM antibodies in serum, plasma, or whole blood, the WHO does not recommend the use of these tests outside of research settings as they have not been validated as yet.[508]
Evidence is particularly weak for point-of-care serological tests, and available evidence does not support their use.
A meta-analysis found that the overall sensitivity of chemiluminescent immunoassays (CLIAs) for IgG or IgM was approximately 98%, and the sensitivity of enzyme-linked immunosorbent assays (ELISAs) was 84%; however, lateral flow immunoassays (LFIAs), which have been developed as point-of-care tests, had the lowest sensitivity at 66%. Test sensitivity was highest 3 or more weeks after onset of symptoms.[509]
Molecular testing (e.g., RT-PCR) is an aid to diagnosis only.
The World Health Organization (WHO) recommends that healthcare providers consider a positive or negative result in combination with specimen type, clinical observations, patient history, and epidemiological information. It also recommends that laboratories ensure that specimens with high cycle threshold values are not incorrectly assigned a positive result due to background noise, and that they provide the cycle threshold value in the report to the healthcare provider. Disease prevalence alters the predictive value of test results. As disease prevalence decreases, the risk of a false positive increases. This means that the probability that a person who has a positive result is truly infected decreases as prevalence decreases, irrespective of the claimed specificity of the test. Careful interpretation of weak positive results is needed.[510]
Interpret RT-PCR test results with caution.
The evidence for the use of RT-PCR in the diagnosis of COVID-19 is limited, and uncertainties about its efficacy and accuracy remain. Estimates of diagnostic accuracy need to be interpreted with caution in the absence of a definitive reference standard to diagnose or rule out COVID-19. Also, more evidence is needed about the efficacy of testing outside of hospital settings and in asymptomatic or mild cases.[502]
Few studies have attempted to culture live SARS-CoV-2 virus from human samples. This is an issue because viral culture is regarded as a recommended test against which any diagnostic index test for viruses must be measured and calibrated, to understand the predictive properties of that test.[511] Prospective routine testing of reference and viral culture specimens is necessary to establish the usefulness and reliability of RT-PCR to diagnose COVID-19, and its relation to patients factors such as date of onset of symptoms and copy threshold, in order to help predict infectivity.[512]
Results can fluctuate from positive to negative at all stages of infection, can become positive again after two consecutive negative tests, can be detected for longer in those with severe infection, and may fluctuate at the level of detection for several weeks. Results may also vary according to the sample site.[58]
It is not clear whether a positive RT-PCR result always indicates the presence of infectious virus.
RT-PCR may overestimate the duration of infectiousness.[58] RT-PCR detects viral RNA, but it is not fully understood how that represents infectious virus. Complete live viruses are necessary for transmission, not the fragments identified by PCR.[512] This could ultimately lead to restrictions for people who do not present an infection risk. Because inactivated RNA degrades slowly over time, it may still be detected many weeks after the patient is no longer infectious.[511]
One study found that only 28.9% of positive RT-PCR SARS-CoV-2 samples demonstrated viral growth when incubated on Vero cells. There was no growth in samples with an RT-PCR cycle threshold >24, or when the symptom onset to test time was >8 days. Therefore, infectivity of patients with a cycle threshold >24 and duration of symptoms >8 days may be low.[513] Another study found that patients with a cycle threshold of 34 or above do not excrete infectious virus.[514] A systematic review found that cycle threshold values were significantly lower and log copies higher in specimens that produce live virus culture. Those with high cycle threshold are unlikely to have infectious potential.[512]
Cycle threshold values do not provide a reliable or consistent proxy for infectiousness across SARS-CoV-2 variants.[515]
Interpreting RT-PCR test results depends on the accuracy of the test itself, and the pre- and post-test probabilities of disease. The accuracy of the result depends on various factors including the site and quality of sampling, stage of disease, degree of viral multiplication or clearance, and disease prevalence.[516]
Sensitivity and specificity: the pooled sensitivity has been estimated to be 87.8%, with the specificity estimated to be in the range of 87.7% to 100%.[502]
Pretest probability: the pretest probability estimate should be made using knowledge of local rates of infection from national and regional data, as well as the patient’s symptoms, potential exposure to cases, a previous medical history of COVID-19 or the presence of antibodies, and the likelihood of an alternative diagnosis.[516] When the pretest probability is low, positive results should be interpreted with caution, and ideally a second specimen tested for confirmation.[517]
Post-test probability: the lower the prevalence of disease in a given population, the lower the post-test probability.[518] For example, if a test with a specificity of 99% is used to test a high-risk symptomatic population where the likelihood of infection is 50%, the positive predictive value is 99%. This means that for every 100 people with a positive test result, 99 people will have SARS-CoV-2 infection but 1 person without infection will have a false-positive result. Conversely, in a low-risk asymptomatic population where the likelihood of infection is low (e.g., 0.05%), the positive predictive value is around 4.3%. This means that for every 100 people with a positive test result, 4 to 5 people will have SARS-CoV-2 infection, but 95 to 96 people without infection will have a false-positive result.[519]
BMJ practice pointer: interpreting a covid-19 test result Opens in new window
False-positive RT-PCR results
False-positive results can be caused by a laboratory error or a cross-reaction with antibodies formed by current and past exposure to seasonal human coronavirus infections (e.g., common cold).[520] False-positive results are more likely when the prevalence of SARS-COV-2 is moderate to low.[521]
There is a lack of data on the rate of false-positive tests. However, preliminary estimates in the UK are in the range of 0.8% to 4%.[522] This rate could translate into a significant proportion of daily false-positive results due to the current low prevalence of the virus in the UK population, adversely affecting the positive predictive value of the test.[517]
Examples of the potential consequences of false-positive test results include:[517]
Unnecessarily postponing or cancelling elective procedures or treatments
Potential exposure to infection following a wrong pathway in hospital settings during urgent hospital admissions
Financial losses due to self-isolation, income losses, and cancelled travel
Psychological damage due to misdiagnosis including fear of infecting others or stigmatisation
Increased depression or domestic violence due to lockdown and isolation
Overestimating the incidence and extent of asymptomatic infection in the population
False-negative RT-PCR results
The US Food and Drug Administration (FDA) has warned that false-negative results may occur with any molecular test for the detection of SARS-CoV-2 if a mutation occurs in the part of the virus’ genome assessed by that test. Multiple genetic targets to determine a final result are less likely to be impacted by increased prevalence of genetic variants. Consider negative results in combination with clinical observations, patient history, and epidemiological information.[523]
False-negative rates of between 2% and 29% have been reported.[516] A systematic review found that the false-negative rate varied across studies from 1.8% to 58% (median 11%); however, there was substantial and largely unexplained heterogeneity across studies.[524]
The probability of a false-negative result in an infected person decreases from 100% on day 1 of infection to 67% on day 4. The median false-negative rate drops to 38% on the day of symptom onset, decreases to 20% on day 8, and then starts to increase again from day 9.[525]
Examples of the potential consequences of false-negative test results include:[516]
Patients may be moved into non-COVID-19 wards leading to spread of hospital-acquired infection.
Carers could spread infection to vulnerable dependents.
Healthcare workers risk spreading the infection to multiple vulnerable individuals.
Evidence for the diagnostic performance of different RT-PCR specimen types is limited.
A systematic review and meta-analysis found that pooled nasal and throat swabs offered the best diagnostic performance of alternative sampling approaches compared with nasopharyngeal swabs for diagnosis in an ambulatory care setting. The sensitivity was 97%, the specificity was 99%, the positive predictive value was 97%, and the negative predictive value was 99%. Throat swabs gave a much lower sensitivity and positive predictive value. Self-collection was not associated with any impairment of diagnostic accuracy.[526] Anterior nasal swabs appear to be less sensitive (82% to 88%) compared with nasopharyngeal swabs (98%). Mid-turbinate and anterior nares swabs perform similarly.[527] There was a high concordance between test results from self-swabbing and assisted swabbing.[528]
Meta-analyses of paired saliva samples and nasopharyngeal swabs found no statistically significant difference in sensitivity or specificity between these specimens for SARS-CoV-2 detection, especially in the ambulatory setting. Sensitivity was not significantly different among asymptomatic people and outpatients. Methods of saliva collection may affect sensitivity. Meta-analyses demonstrate that saliva is as valid as nasopharyngeal sampling for the detection of SARS-CoV-2 infections in symptomatic and asymptomatic patients. Saliva sampling is simple, fast, non-invasive, inexpensive, and painless.[529][530][531][532][533][534]
A Cochrane review found that self-collected samples from the nose, oropharynx, oral cavity, and saliva were less sensitive for detecting COVID-19 compared with healthcare worker-collected nasopharyngeal samples. However, self-collected gargle and deep throat saliva samples had similar sensitivity compared with healthcare worker-collected nasopharyngeal samples.[535]
Rapid antigen tests are more reliable in people with a high viral load who are symptomatic.
Rapid antigen testing appears to be a reliable diagnostic tool to quickly detect people with a high viral load and in the first week of symptom onset, and can help to detect and isolate potential superspreaders before RT-PCR results are available. However, testing is unsuccessful in detecting people with lower viral load and asymptomatic patients.[536][537]
A Cochrane review found that rapid antigen tests vary in sensitivity. Sensitivity was higher in the first week after symptom onset in symptomatic people (80.9%) compared with the second week of symptoms (53.8%). Sensitivity was higher in symptomatic people (73%) compared with asymptomatic people (54.7%). Sensitivity also varied between brands of tests.[538]
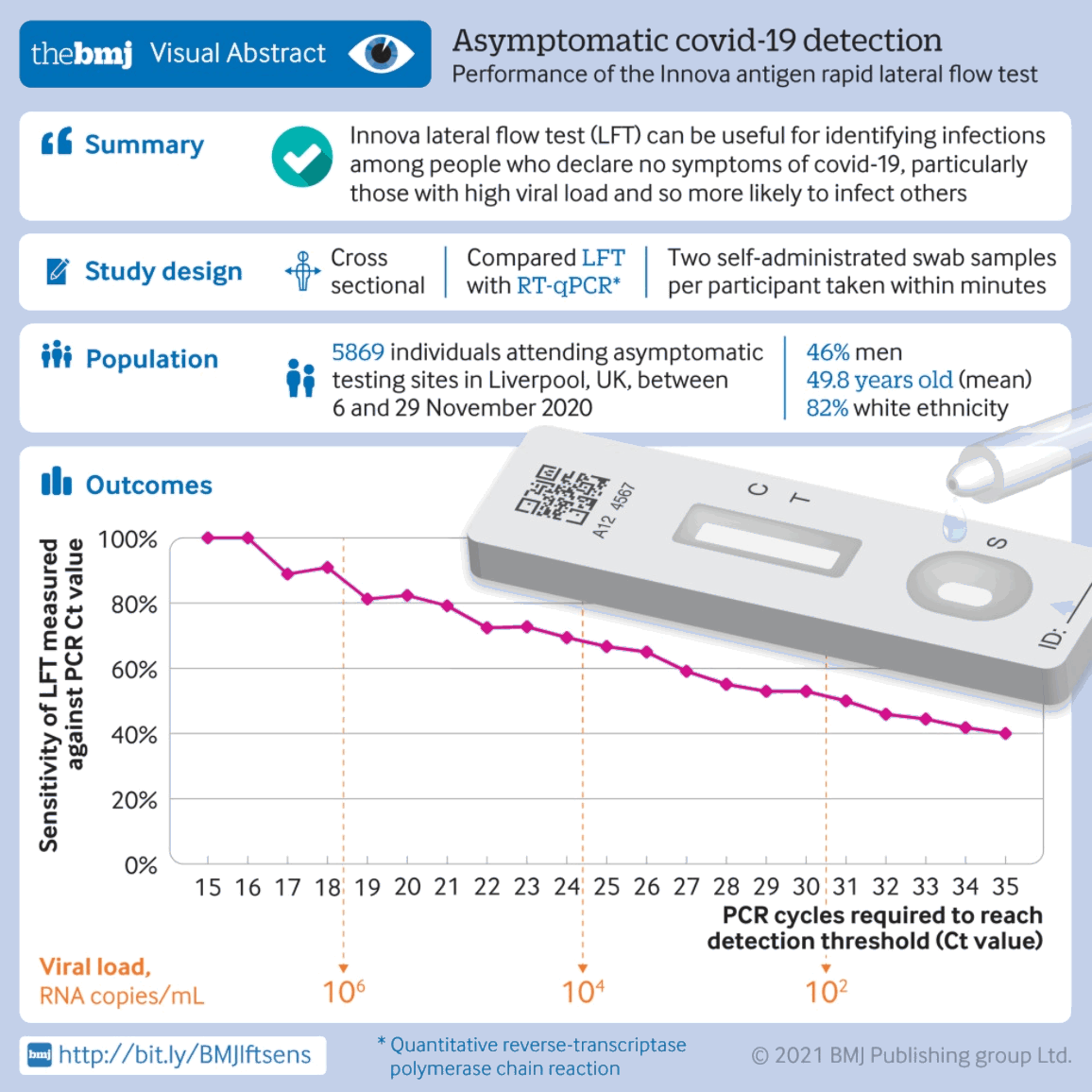
An observational cohort study that assessed the performance of rapid antigen lateral flow testing against RT-PCR in an asymptomatic general population in the UK found that the lateral flow test can be useful for detecting infections among asymptomatic adults, particularly those with a high viral load who are likely to be infectious. Lateral flow tests showed a sensitivity of 40%, specificity of 99.9%, positive predictive value of 90.3%, and negative predictive value of 99.2% in this population. Approximately 10% of people with a higher viral load detected by RT-PCR were missed by lateral flow tests.[539][Figure caption and citation for the preceding image starts]: Performance of the Innova SARS-CoV-2 antigen rapid lateral flow testBMJ. 2021;374:n1637 [Citation ends].
A systematic review and meta-analysis found that fully self-performed rapid antigen tests, including self-collection of nasopharyngeal or oropharyngeal samples, showed moderate sensitivity and high specificity compared with standard RT-PCR tests. The sensitivity was higher in symptomatic patients when samples were collected within 7 days of symptom onset.[540]
A Cochrane review found that self-collected samples from the nose have a similar sensitivity to healthcare worker-collected nasopharyngeal samples.[535]
Results should be interpreted with caution as false-positive and false-negative results are possible.
The FDA has warned that false-positive results can occur with rapid antigen tests, including when users do not follow the instructions for use, and that the number of false-positive tests increases as disease prevalence decreases.[541]
The FDA recommends repeat (or serial) testing following a negative result from an at-home test, regardless of whether symptoms are present, in order to reduce the risk of a false-negative result.[542]
Certain tests may not be recommended due to performance issues.
Serological testing should be used with caution and not be used to determine acute infections.[439]
A reliable diagnosis is often only possible in the recovery phase when opportunities for management or interruption of transmission have passed.[439]
The presence of antibodies that bind to SARS-CoV-2 does not guarantee that they are neutralising antibodies, or that they offer protective immunity.[439]
Evidence for the use of serological testing is limited and uncertainties about its accuracy remain.
A Cochrane review found that antibody tests could be a useful tool for patients in whom molecular- or antigen-based tests have failed to detect SARS-CoV-2 virus, including in those with ongoing symptoms of acute infection (from week 3 onwards) or those presenting with post-acute sequelae. Test sensitivity is too low during the first week since the onset of symptoms.[543]
The estimated sensitivity of antibody tests ranged from 18.4% to 96.1% (the lowest reported sensitivity was from a point-of-care test, although a sensitivity <50% was reported for one laboratory test), and specificity ranged from 88.9% to 100%. More evidence is needed about the efficacy of testing outside of hospital settings and in asymptomatic or mild cases.[502]
Imaging should be used as one element of the diagnostic work-up that otherwise includes clinical and laboratory data.[544]
Choice of imaging modality depends on various factors and you should consult your local protocols.
All imaging procedures should be performed according to local infection prevention and control procedures.
Chest imaging is considered safe in pregnant women.[545]
Order a chest x-ray in all patients who are seriously ill (e.g., SpO2 <94% or National Early Warning Score 2 [NEWS2] score ≥3) or those who are stable but a chest x-ray is clinically indicated (e.g., suspected pneumonia).[443]
Approximately 74% of patients have an abnormal chest x-ray at the time of diagnosis. The most common abnormalities are ground-glass opacity (29%) and consolidation (28%). Distribution is generally bilateral, peripheral, and basal zone predominant. Pneumothorax and pleural effusions are rare. There is no single feature on chest x-ray that is diagnostic.[546]
Chest x‐ray is moderately sensitive and moderately specific for the diagnosis of COVID‐19. Pooled results found that chest x‐ray correctly diagnosed COVID‐19 in 73% of people who had the disease. However, it incorrectly identified COVID‐19 in 27% of people who did not have the disease.[547]
Although chest x-ray appears to have a lower sensitivity compared with chest CT, it has the advantages of being less resource-intensive, associated with lower radiation doses, easier to repeat sequentially, and portable.[544]
Consider ordering a CT scan of the chest.
Chest CT may play a role in diagnosis in a limited number of hospitalised patients, particularly when initial molecular testing has been inconclusive, or when an alternative diagnosis is being considered.[548] However, it is not diagnostic for COVID-19 and local guidance should be consulted on whether to perform a CT scan.
The British Society of Thoracic Imaging (BSTI) recommends CT imaging in patients with clinically suspected COVID-19 who are seriously ill if chest x-ray is uncertain or normal. Without the suspicion of COVID-19, the radiology is non-specific and could represent many other disease processes. The BSTI in collaboration with NHS England have produced a radiology decision support tool to help clinicians decide whether or not chest imaging should be ordered.[443] BSTI: radiology decision tool for suspected COVID-19 Opens in new window
Some institutions in the UK recommend a more pragmatic approach for patients with high clinical suspicion of COVID-19, with chest CT recommended only after two indeterminate or normal chest x-rays in combination with a negative RT-PCR test.[549]
The American College of Radiology recommends reserving CT for hospitalised, symptomatic patients with specific clinical indications for CT, and emphasises that a normal chest CT does not mean that a patient does not have COVID-19 and that an abnormal chest CT is not specific for COVID-19 diagnosis.[550]
Chest CT is moderately sensitive (70%) and highly specific (90%) for the diagnosis of COVID‐19.[551]
Pooled results found that chest CT correctly diagnosed COVID‐19 in 87% of people who had the disease. However, it incorrectly identified COVID‐19 in 21% of people who did not have the disease. Therefore, chest CT may have more utility for excluding COVID‐19 than for differentiating it from other causes of respiratory illness.[547]
The positive predictive value was low (1.5% to 30.7%) in low-prevalence regions, and the negative predictive value ranged from 95.4% to 99.8% in one meta-analysis. Pooled sensitivity and specificity were 94% to 96% and 37%, respectively.[552][553]
Accuracy appears to be lower among children; however, there are limited data in this population.[548]
Chest CT results may not correlate with RT-PCR test results or clinical presentation.
Evidence of pneumonia on CT may precede a positive RT-PCR result for SARS-CoV-2 in some patients.[554]
Some patients may present with a normal chest finding despite a positive RT-PCR.[555]
Results of RT-PCR testing may be false-negative, so patients with typical CT findings should have repeat RT-PCR testing to confirm the diagnosis.[556]
CT imaging abnormalities may be present in asymptomatic patients. The pooled estimate of the rate of positive chest CT findings in asymptomatic cases was 47.6% (mainly ground-glass opacity).[557]
Patients with Omicron variants of SARS-CoV-2 were less likely to be CT-positive compared with those infected with non-Omicron variants.[558]
Typical features of chest CT include ground-glass opacity and consolidation.
Abnormal chest CT findings have been reported in up to 97% of hospitalised patients.[559]
The most common findings are ground-glass opacity, either in isolation or co-existing with other findings such as consolidation, interlobular septal thickening, or crazy-paving pattern. The most common distribution pattern is bilateral, peripheral/subpleural, posterior distribution of the opacities, with a lower lobe predominance. Extensive/multilobar involvement with consolidations is more common in older patients and those with severe disease.[560]
Atypical features include pulmonary vascular enlargement, adjacent pleural thickening, air bronchograms, subpleural lines, bronchus distortion, bronchiectasis, vacuolar retraction sign, and halo sign. Pleural effusion, pericardial effusion, cavitation, pneumothorax, and mediastinal lymphadenopathy have been reported rarely.[560]
Ground-glass opacity has the highest diagnostic performance for COVID-19 pneumonia, followed by ground-glass opacity plus consolidation, and consolidation only.[561]
The simultaneous presence of ground-glass opacity and other features of viral pneumonia had optimum performance in the detection of COVID-19 (sensitivity 90% and specificity 89%).[562]
CT scan generally shows an increase in the size, number, and density of ground-glass opacities in the early follow-up period, with a progression to mixed areas of ground-glass opacities, consolidations, and crazy paving peaking at day 10 to 11, before gradually resolving or persisting as patchy fibrosis.[560]
A small comparative study found that patients with COVID-19 are more likely to have bilateral involvement with multiple mottling and ground-glass opacity compared with other types of pneumonia.[563]
Patients with non-Omicron variants of SARS-CoV-2 had more imaging changes on CT compared with those with Omicron variants.[558]
Patients with Omicron variants typically had lesions located in the centre of the lung or in a single lobe. They were less likely to have bilateral lesions, crazy-paving pattern, linear opacity, and vascular enlargement, but were more likely to have bronchial wall thickening. The distribution of lesions was more extensive (bilateral and multilobe involvement) in patients with non-Omicron variants.
Pregnant women appear to present more commonly with more advanced CT findings compared with the general adult population.[564]
However, CT results are similar to those in the general adult population.
Children frequently have normal or mild CT chest findings.
The most common signs in children are patchy ground-glass opacity, non-specific patchy shadows, areas of consolidation, infected nodules, and a halo sign. Abnormalities are more common in multiple lobes and are predominantly bilateral. Pleural effusion is rare. Children may have signs of pneumonia on chest imaging despite having minimal or no symptoms.[565]
Ground-glass opacity and peribronchial thickening were the most prevalent findings in infants younger than 1 year of age.[566]
Lung ultrasound may be an alternative to chest x-ray and chest CT in some locations.
Lung ultrasound is used as a diagnostic tool in some centres as an alternative to chest x-ray and chest CT.
Although there is only very low-certainty evidence supporting its diagnostic accuracy, it might be helpful as a supplemental or alternate imaging modality.[544]
BSTI: lung ultrasound (LUS) for COVID-19 patients in critical care areas Opens in new window
Lung ultrasound is sensitive but not specific for the diagnosis of COVID‐19.
Pooled results found that lung ultrasound correctly diagnosed COVID‐19 in 87% of people with the disease. However, it incorrectly diagnosed COVID‐19 in 24% of people who did not have the disease. Therefore, ultrasound may have more utility for excluding COVID‐19 than for differentiating it from other causes of respiratory illness.[547]
Lung ultrasound score was a good predictor of poor outcome and 28-day mortality in one study.[567]
Typical features include B-lines (confluent or separated and usually at least 3) and pleural abnormalities, with a bilateral distribution.[568]
Other findings include consolidations, pleural effusion, air bronchogram, and pneumothorax.
While these findings are not specific for COVID-19, they may increase the likelihood of disease in the context of a characteristic clinical presentation.
Lung ultrasound has advantages and limitations.[569][570][571]
Advantages: portability; bedside evaluation; reduced healthcare worker exposure; easier sterilisation process; absence of ionising radiation exposure; repeatability during follow-up; safe to use in pregnant women and children; more readily available in resource-limited settings.
Limitations: unable to discern chronicity of a lesion; other imaging modalities may be required.
Possible roles for ultrasound include: reducing nosocomial transmission; monitoring progress of patients; and a possible role in subpopulations who are vulnerable but are not suitable for CT (e.g., pregnant women).[572] Lung ultrasound score may play a role in prognosis.[573]
The World Health Organization (WHO) recommends chest imaging (chest x-ray, chest CT, or lung ultrasound) in the following scenarios:[544]
Symptomatic patients with suspected COVID-19 when RT-PCR is not available, RT-PCR test results are delayed, or initial RT-PCR testing is negative but there is a high clinical suspicion for COVID-19 (for diagnosis)
Patients with suspected or confirmed COVID-19 who are not currently hospitalised and have mild symptoms (to decide on hospital admission versus home discharge)
Patients with suspected or confirmed COVID-19 who are not currently hospitalised and have moderate to severe symptoms (to help decide on regular ward admission versus intensive care unit admission)
Patients with suspected or confirmed COVID-19 who are currently hospitalised and have moderate to severe symptoms (to inform therapeutic management).
Использование этого контента попадает под действие нашего заявления об отказе от ответственности