Summary
Definition
History and exam
Key diagnostic factors
- cough with increasing sputum production
- fever or chills
- dyspnea
- pleuritic pain
- abnormal auscultatory findings
Risk factors
- age >65 years
- residence in a healthcare setting
- COPD
- exposure to cigarette smoke
- alcohol abuse
- poor oral hygiene
- use of acid-reducing drugs, inhaled corticosteroids, antipsychotics, antidiabetic drugs
- contact with children
- HIV infection
- diabetes mellitus
- chronic renal disease
- chronic liver disease
- use of opioids
Diagnostic tests
1st tests to order
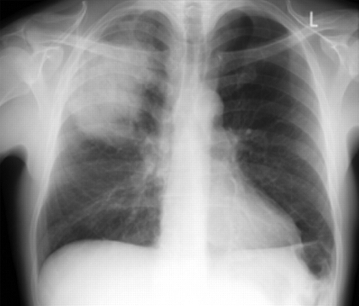
- chest x-ray
- CBC
- serum electrolytes/blood urea nitrogen
- liver function tests
- blood glucose
- arterial blood gases/oximetry
- blood culture
- sputum culture
Tests to consider
- point-of-care lung ultrasound
- CT chest
- urinary antigen testing for Legionella and pneumococcus
- serum C-reactive protein
- serum procalcitonin
- thoracocentesis and pleural fluid culture
- bronchoscopy
- tests for respiratory viruses
- molecular microbiological techniques
Treatment algorithm
Contributors
Expert advisers
Catia Cilloniz, MD, PhD, FERS
Applied Research in Respiratory Diseases
Hospital Clinic of Barcelona
CIBERES
IDIBAPS
Associate Professor
University of Barcelona
Barcelona
Spain
Disclosures
CC is an author of a number of references cited in this topic.
Antoni Torres, MD, PhD
Professor of Medicine
Director
Pulmonary Intensive Care Unit
Respiratory Institute
Hospital Clinic of Barcelona
Barcelona
Spain
Disclosures
AT is an author of a number of references cited in this topic.
Peer reviewers
Barbara Jones, MD, MSc
Assistant Professor
Division of Pulmonary and Critical Care Medicine
University of Utah
Salt Lake City
UT
Disclosures
BJ declares that she has no competing interests.
Denise Nassisi, MD
Associate Professor
Departments of Emergency Medicine and Medicine
Icahn School of Medicine at Mount Sinai
New York
NY
Disclosures
DN declares that she has no competing interests.
Jeremy Brown, MBBS, FRCP, PhD
Professor of Respiratory Infection/Honorary Consultant
University College London
London
UK
Disclosures
JB was a member of the NICE pneumonia guidelines committee.
Grant Waterer, MBBS, PhD, MBA, FRACP, FCCP
Professor of Medicine
Royal Perth Hospital
Perth
Australia
Disclosures
GW declares that he has no competing interests.
Nathan Dean, MD
Professor (clinical) of Medicine
Section Chief Pulmonary Critical Care Medicine
University of Utah
Intermountain Medical Center
Murray
UT
Disclosures
ND declares that he has no competing interests.
Peer reviewer acknowledgements
BMJ Best Practice topics are updated on a rolling basis in line with developments in evidence and guidance. The peer reviewers listed here have reviewed the content at least once during the history of the topic.
Disclosures
Peer reviewer affiliations and disclosures pertain to the time of the review.
References
Key articles
Metlay JP, Waterer GW, Long AC, et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019 Oct 1;200(7):e45-67.Full text Abstract
Reference articles
A full list of sources referenced in this topic is available here.
Use of this content is subject to our disclaimer