Resumo
Definição
História e exame físico
Principais fatores diagnósticos
- presença de fatores de risco
Outros fatores diagnósticos
- sintomas constitucionais
- icterícia
- ascite
- sinais de encefalopatia hepática
- manifestações extra-hepáticas
Fatores de risco
- práticas clínicas não seguras
- uso de drogas por via intravenosa ou intranasal
- transfusão de sangue ou transplante de órgão
- consumo intenso de bebidas alcoólicas
- polimorfismo no gene interleucina (IL) 28B
- vírus da imunodeficiência humana (HIV)
- prisão/institucionalização
- diálise
- trabalho na área da saúde
- tatuagens/piercings corporais
- acupuntura
- vários parceiros sexuais
- mãe infectada (para o feto)
- sexo masculino
- deficiência de vitamina D
Investigações diagnósticas
Primeiras investigações a serem solicitadas
- ensaio imunoenzimático (EIE) para anticorpos contra o vírus da hepatite C (HCV)
- reação em cadeia da polimerase para RNA de vírus da hepatite C (HCV)
- aminotransferases séricas
Investigações a serem consideradas
- genotipagem viral
- ensaio de antígeno de núcleo do vírus da hepatite C (HCV)
- testes não invasivos de fibrose hepática ou elasticidade
- índice da relação aspartato aminotransferase sobre plaquetas (APRI)
- biópsia hepática
- testes para coinfecção
Algoritmo de tratamento
infecção recente
infecção crônica adequada para tratamento simplificado: qualquer genótipo
infecção crônica não adequada para tratamento simplificado ou pacientes virgens de tratamento: genótipo 1a
infecção crônica não adequada para tratamento simplificado ou pacientes virgens de tratamento: genótipo 1b
infecção crônica não adequada para tratamento simplificado ou pacientes virgens de tratamento: genótipo 2
infecção crônica não adequada para tratamento simplificado ou pacientes virgens de tratamento: genótipo 3
infecção crônica não adequada para tratamento simplificado ou pacientes virgens de tratamento: genótipo 4
infecção crônica não adequada para tratamento simplificado ou pacientes virgens de tratamento: genótipo 5 ou 6
infecção crônica não adequada para tratamento simplificado ou pacientes com tratamento prévio: todos os genótipos
cirrose descompensada
Colaboradores
Autores
Jawad Ahmad, MD, FRCP, FAASLD
Professor of Medicine
Division of Liver Diseases
Mount Sinai Hospital
New York
NY
Declarações
JA declares that he has no competing interests.
Agradecimentos
Dr Jawad Ahmad would like to gratefully acknowledge Dr Brian L. Pearlman, the previous contributor to this topic.
Declarações
BLP is on the speakers' bureaus and serves as an advisor to Merck, Gilead, J&J, and AbbVie, and does contracted research with Boehringer Ingelheim, Tibotec/Janssen, Bristol-Myers Squibb, Gilead, and Merck. BLP is also an author of a number of references cited in this topic. BLP wishes to acknowledge Chaithanya Mallikarjun, MD, for her help in writing the original version of the manuscript.
Revisores
AnnMarie Liapakis, MD
Assistant Professor
Digestive Disease and Liver Transplant
Yale University
CT
Declarações
AL has participated in an advisory board meeting with Gilead and Janssen, and is a primary investigator for Merck’s C-Surfer trial.
Benedict Maliakkal, MRCP
Director of Hepatology and Medical Director of Liver Transplantation
Associate Professor of Medicine
Strong Memorial Hospital
University of Rochester
NY
Declarações
BM belongs to the paid speaker’s bureau for pharmaceutical companies Gilead (maker of ledipasvir/sofosbuvir), AbbVie (maker of Viekira Pak® - ombitasvir/paritaprevir/ritonavir and dasabuvir), Merck, and Salix.
Sulleman Moreea, FRCS (Glasg), FRCP
Consultant Hepatologist
Bradford Teaching Hospitals Foundation Trust
Bradford
UK
Declarações
SM has received sponsorship and has sat on advisory boards for BMS, Gilead, AbbVie, and Merck.
Steve Ryder, DM, FRCP
Consultant Hepatologist
Nottingham University Hospitals NHS Trust
UK
Declarações
SR is an advisory board (paid) member for AbbVie, Gilead, Janssen, and MSD.
Créditos aos pareceristas
Os tópicos do BMJ Best Practice são constantemente atualizados, seguindo os desenvolvimentos das evidências e das diretrizes. Os pareceristas aqui listados revisaram o conteúdo pelo menos uma vez durante a história do tópico.
Declarações
As afiliações e declarações dos pareceristas referem--se ao momento da revisão.
Referências
Principais artigos
American Association for the Study of Liver Diseases; Infectious Diseases Society of America. HCV guidance: recommendations for testing, managing, and treating hepatitis C. Dec 2023 [internet publication].Texto completo
Artigos de referência
Uma lista completa das fontes referenciadas neste tópico está disponível para os usuários com acesso total ao BMJ Best Practice.
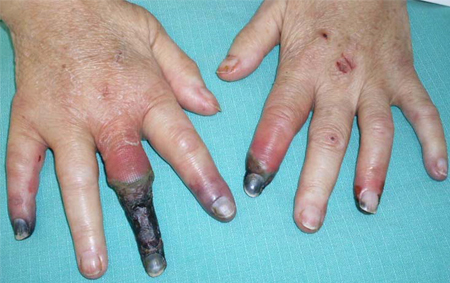
Diagnósticos diferenciais
- Hepatite B
- Hepatite D
- Hepatite E
Mais Diagnósticos diferenciaisDiretrizes
- Clinical screening and diagnosis for hepatitis C
- Bloodborne viruses in healthcare workers: health clearance and management
Mais DiretrizesFolhetos informativos para os pacientes
Hepatite C: perguntas a fazer ao seu médico
Hepatite C: o que é?
Mais Folhetos informativos para os pacientesConectar-se ou assinar para acessar todo o BMJ Best Practice
O uso deste conteúdo está sujeito ao nosso aviso legal