Resumen
Definición
Anamnesis y examen
Principales factores de diagnóstico
- presencia de factores de riesgo
- despigmentación de la piel de la zona acral y perioral
- despigmentación de la piel de la zona perianal y genital
- traumatismo cutáneo reciente
- dolor localizado por quemadura solar
- realce y fluorescencia con exposición a radiación UVA
- nevo con halo
- despigmentación universal
Factores de riesgo
- edad <30 años
- antecedentes familiares de vitiligo
- enfermedad autoinmune
- contacto químico
Pruebas diagnósticas
Primeras pruebas diagnósticas para solicitar
- diagnóstico clínico
Pruebas diagnósticas que deben considerarse
- Examen con lámpara de Wood
- biopsia de piel
Algoritmo de tratamiento
vitiligo segmentario o vitiligo limitado
vitiligo generalizado (más del 3% del área de superficie corporal)
Colaboradores
Autores
John E. Harris, MD, PhD
Professor
Department of Dermatology
University of Massachusetts Medical School
Worcester
MA
Divulgaciones
JEH has acted as a consultant and/or investigator for: Pfizer, Genzyme/Sanofi, Aclaris Therapeutics Inc., Incyte, Rheos Medicines, Sun Pharmaceuticals, LEO Pharma, Villaris Therapeutics Inc., Dermavant, Temprian, AbbVie Inc., Janssen, TeVido, BioDevices, EMD Serono, Almirall, Boston Pharma, Sonoma Biotherapeutics Inc., Methuselah Health, Twi Biotech, Pandion, Cogen Therapeutics Inc., Admirx, BridgeBio, AnaptysBio, Avita, Frazier Management. He has equity in TeVido Biodevices, Rheos, and Villaris Therapeutics Inc., and is the Scientific Founder of Villaris Therapeutics Inc. JEH is an author of a number of references cited in this topic.
Mehdi Rashighi, MD
Assistant Professor
Department of Dermatology
University of Massachusetts Medical School
Worcester
MA
Divulgaciones
MR has received consulting fees and/or research grants from the following: Pfizer, AbbVie, Dermavant, LEO Pharma, and Almirall. MR is an author of a reference cited in this topic.
Agradecimientos
Professor John E. Harris and Dr Mehdi Rashighi would like to gratefully acknowledge Dr Bernhard Ortel, a previous contributor to this topic. BO declares that he has no competing interests.
Revisores por pares
Iltefat Hamzavi, MD
Senior Staff Physician
Multicultural Dermatology Center
Henry Ford Hospital
Detroit
MI
Divulgaciones
IH is an author of a number of references cited in this topic.
Piergiacomo Calzavara-Pinton, MD
Chair
Dermatology Department
University of Brescia
Brescia
Italy
Divulgaciones
PC-P is an author of a number of references cited in this topic.
Agradecimiento de los revisores por pares
Los temas de BMJ Best Practice se actualizan de forma continua de acuerdo con los desarrollos en la evidencia y en las guías. Los revisores por pares listados aquí han revisado el contenido al menos una vez durante la historia del tema.
Divulgaciones
Las afiliaciones y divulgaciones de los revisores por pares se refieren al momento de la revisión.
Referencias
Artículos principales
Ortonne J-P. Vitiligo and other disorders of hypopigmentation. In: Bolognia JL, Jorrizzo JL, Rapini RP, eds. Dermatology. New York, NY: Mosby; 2008:913-38.
Taieb A, Picardo M. Clinical practice: vitiligo. N Engl J Med. 2009 Jan 8;360(2):160-9. Resumen
Taieb A, Alomar A, Böhm M, et al; Vitiligo European Task Force (VETF); European Academy of Dermatology and Venereology (EADV); Union Européenne des Médecins Spécialistes (UEMS). Guidelines for the management of vitiligo: the European Dermatology Forum consensus. Br J Dermatol. 2013 Jan;168(1):5-19.Texto completo Resumen
Whitton ME, Pinart M, Batchelor J, et al. Interventions for vitiligo. Cochrane Database Syst Rev. 2015 Feb 24;(2):CD003263.Texto completo Resumen
Artículos de referencia
Una lista completa de las fuentes a las que se hace referencia en este tema está disponible para los usuarios con acceso a todo BMJ Best Practice.
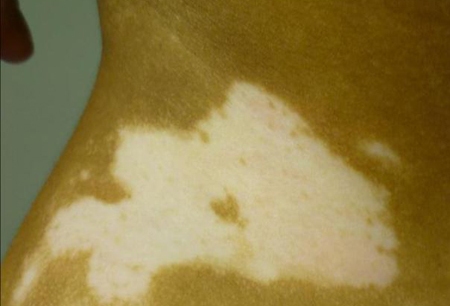
Diferenciales
- Piebaldismo
- Síndrome de Waardenburg
- Esclerosis tuberosa
Más DiferencialesGuías de práctica clínica
- British Association of Dermatologists guidelines for the management of people with vitiligo 2021
- Guidelines for the management of vitiligo
Más Guías de práctica clínicaInicie sesión o suscríbase para acceder a todo el BMJ Best Practice
El uso de este contenido está sujeto a nuestra cláusula de exención de responsabilidad
