Evaluation of cranial nerve mononeuropathy
- Overview
- Theory
- Emergencies
- Diagnosis
- Resources
Summary
Cranial nerve mononeuropathies present with varying signs and symptoms depending on which nerve is affected. The effects of the mononeuropathy also depend on where in its pathway the nerve is affected, and the etiology.
There are 12 paired cranial nerves, named and numbered according to the rostral-caudal order of attachment to the brain. They serve a variety of functions and predominantly provide the motor and sensory innervation to the head.
[Figure caption and citation for the preceding image starts]: The 12 cranial nerves and their functional end organsAdapted from An Introduction to Neurophysiology, Thomy Nilsson (2022). Reproduced under the Creative Commons CC BY-SA 4.0 license (https://creativecommons.org/licenses/by-sa/4.0/) [Citation ends].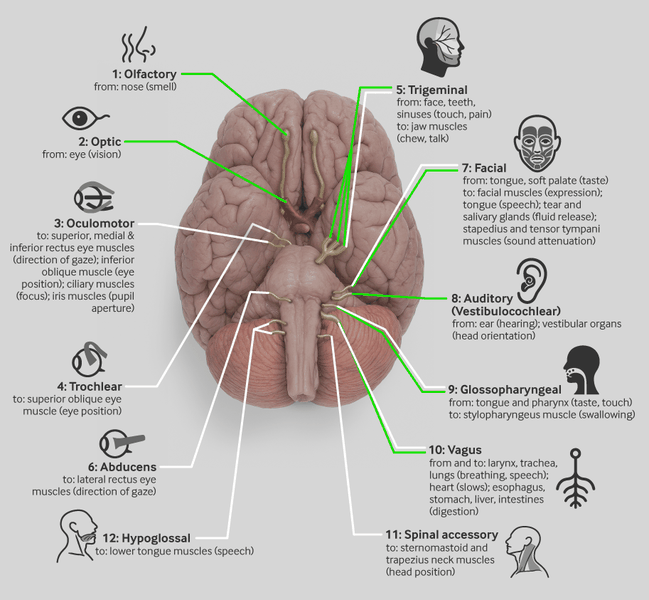
Olfactory (I)
Anatomy
Olfaction begins with transduction of odorants from the air into the nasal mucosa. These odorants diffuse or are transported to bipolar receptor cells located in the olfactory neuroepithelium in the roof of the nasal chamber. Action potentials are induced in these cells, which synapse with olfactory bulb glomeruli.[1]Moran DT, Rowley JC 3rd, Jafek BW, et al. The fine structure of the olfactory mucosa in man. J Neurocytol. 1982;11:721-746. http://www.ncbi.nlm.nih.gov/pubmed/7143026?tool=bestpractice.com The receptor cell axons project through the cribriform plate of the ethmoid bone and synapse within the glomerular layer of the olfactory bulb. The paired olfactory bulbs are located at the base of the frontal lobe overlying the cribriform plate.[2]Doty RL. The olfactory system and its disorders. Semin Neurol. 2009;29:74-81. http://www.ncbi.nlm.nih.gov/pubmed/19214935?tool=bestpractice.com The second-order neurons leave the olfactory bulb to synapse on the primary olfactory cortex. These areas encode characteristics of odor quality, identity, familiarity, and emotion.[3]Gottfried JA, Deichmann R, Winston JS, et al. Functional heterogeneity in human olfactory cortex: an event-related functional magnetic resonance imaging study. J Neurosci. 2002;22:10819-10828. http://www.jneurosci.org/content/22/24/10819.full http://www.ncbi.nlm.nih.gov/pubmed/12486175?tool=bestpractice.com
Function and disorders
Changes in olfactory function frequently go unnoticed and often do not present to a clinician.[2]Doty RL. The olfactory system and its disorders. Semin Neurol. 2009;29:74-81. http://www.ncbi.nlm.nih.gov/pubmed/19214935?tool=bestpractice.com Patients may notice altered taste, rather than a loss of sense of smell.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com Olfaction is critically important for safety, nutritional status, and quality of life. Disorders can manifest as a total loss of smell (anosmia), partial loss of smell (hyposmia), distortions (dysosmias), or spontaneous olfactory hallucinations (phantosmias).[2]Doty RL. The olfactory system and its disorders. Semin Neurol. 2009;29:74-81. http://www.ncbi.nlm.nih.gov/pubmed/19214935?tool=bestpractice.com Infrequently, olfactory dysfunction can be the presenting sign/symptom of neurodegenerative disorders (such as idiopathic Parkinson disease), or an intracranial mass lesion.
Nerve testing
The diagnosis can usually be made clinically. Commercial odor identification tests are available, which require patients to identify several predefined smells.[5]Doty RL, Shaman P, Dann M. Development of the University of Pennsylvania Smell Identification Test: a standardized microencapsulated test of olfactory function. Physiol Behav. 1984;32:489-502. http://www.ncbi.nlm.nih.gov/pubmed/6463130?tool=bestpractice.com These may be useful to confirm olfactory dysfunction. Psychophysical tests are useful to validate and classify olfactory dysfunction, but establishing the cause of olfactory loss relies heavily on the history. Olfactory evoked potentials are available in specialist centers.
Optic (II)
Anatomy
Axons making up the optic nerve arise from retinal ganglion cells. These axons run toward the lamina cribrosa and merge in the optic papilla. At this point, they form the optic nerve. In the orbital apex, the nerve passes through the extraocular muscle origins and enters the optic canal. The nerve continues to course upward and inward until it meets with the contralateral nerve to form the optic chiasm superior to the sella and pituitary gland.[6]Selhorst JB, Chen Y. The optic nerve. Semin Neurol. 2009;29:29-35. http://www.ncbi.nlm.nih.gov/pubmed/19214930?tool=bestpractice.com Action potentials are then carried to the lateral geniculate body. The intraorbital portion is surrounded by the subarachnoid space and dura that extends from the intracranial cavity. The central retinal artery and vein course through the middle of the nerve.
Function and disorders
Humans have a highly developed visual system, which transmits information from the environment. The optic nerve carries millions of fibers from the retina into the central nervous system (CNS).[7]Curcio CA, Allen KA. Topography of ganglion cells in human retina. J Comp Neurol. 1990;300:5-25. http://www.ncbi.nlm.nih.gov/pubmed/2229487?tool=bestpractice.com Vision is critical for human function and, as such, optic nerve pathology can severely affect quality of life.[6]Selhorst JB, Chen Y. The optic nerve. Semin Neurol. 2009;29:29-35. http://www.ncbi.nlm.nih.gov/pubmed/19214930?tool=bestpractice.com Optic nerve lesions typically produce monocular visual loss, which can be sudden or gradual, and may or may not be associated with pain. The potential causes of optic neuropathy are diverse and include vascular, toxic, metabolic, traumatic, compressive, infectious, inflammatory, and idiopathic etiologies.
Nerve testing
Symptoms of optic nerve damage can represent changes in visual acuity, contrast, brightness, or colour.[6]Selhorst JB, Chen Y. The optic nerve. Semin Neurol. 2009;29:29-35. http://www.ncbi.nlm.nih.gov/pubmed/19214930?tool=bestpractice.com A detailed description of visual dysfunction is essential and can narrow the differential. To define the degree of optic nerve dysfunction, the following tests are frequently performed.
Visual acuity: tested using a Snellen chart.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com Optic nerve damage may result in central visual loss.
Color vision: assessed with a series of color plates. Patients with unilateral optic nerve impairment have difficulty identifying colors (dyschromatopsia); color perception is more likely to be significantly affected than visual acuity.
Pupillary testing: pupillary light reflex testing for relative afferent pupillary defect (RAPD) is the only bedside test of optic nerve dysfunction that is independent of patient’s subjective response.[6]Selhorst JB, Chen Y. The optic nerve. Semin Neurol. 2009;29:29-35. http://www.ncbi.nlm.nih.gov/pubmed/19214930?tool=bestpractice.com
Visual fields testing: a basic visual field test can be performed at the bedside by comparing the patient's peripheral vision with the clinician.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com If a defect is identified, formal testing may be required, for example with Goldmann perimetry. The more central part of the visual field may be tested using an Amsler grid.
Direct ophthalmoscopy: visualizing the optic nerve as it enters the back of the eye can reveal pallor (optic atrophy) or disk swelling (papillitis or papilledema).
Electrodiagnostic testing: visual evoked potentials (VEPs) can be performed to objectively assess for conduction slowing at the optic nerve. This potential is recorded from surface electrodes on the scalp while displaying visual patterns or light flashes to either eye. Note that visual pathway lesions posterior to the optic chiasm are technically more challenging to identify with VEP.[8]Rubin DI. Clinical neurophysiology. 5th ed. Oxford: Oxford University Press; 2021.
Oculomotor (III), trochlear (IV), and abducens (VI)
Anatomy
The oculomotor nerve emerges from the midbrain nucleus that lies ventral to the sylvian aqueduct. One unpaired and 4 paired subnuclei can be distinguished. The most dorsal subnucleus contains the visceral Edinger-Westphal nucleus and the levator palpebrae nucleus. The Edinger-Westphal nucleus mediates pupillary constriction. Laterally the dorsal, intermediate, and ventral subnuclei provide innervation to the ipsilateral inferior rectus, inferior oblique, and medial rectus, respectively. The oculomotor nerve fascicles leave the nucleus and pass ventrally through the red nucleus before exiting just medial to the cerebral peduncles. In the subarachnoid space the third nerve passes between the superior cerebellar and posterior cerebral arteries. The nerve then enters the lateral wall of the cavernous sinus and divides into a superior and inferior branch as it enters the orbit through the superior orbital fissure.[9]Brazis PW. Isolated palsies of cranial nerves III, IV, and VI. Semin Neurol. 2009;29:14-28. http://www.ncbi.nlm.nih.gov/pubmed/19214929?tool=bestpractice.com The oculomotor nerve contains inner somatic nerve fibers innervating the extraocular muscles, surrounded by outer autonomic nerve fibers mediating pupillary constriction.[10]Park HK, Rha HK, Lee KJ, et al. Microsurgical anatomy of the oculomotor nerve. Clin Anat. 2017 Jan;30(1):21-31. http://www.ncbi.nlm.nih.gov/pubmed/27859787?tool=bestpractice.com
The trochlear nucleus is located in the midbrain tegmentum at the level of the inferior colliculus. The nerve fascicles course posteroinferiorly to decussate at the anterior medullary velum before exiting from the dorsal aspect of the midbrain. The trochlear nerve is the only nerve to arise from the dorsal aspect of the brainstem. The fourth nerve traverses the brainstem cisterns close to the undersurface of the tentorial edge and pierces the dura to enter the lateral cavernous sinus. The trochlear nerve enters the orbit through the superior orbital fissure to innervate the superior oblique muscle.[9]Brazis PW. Isolated palsies of cranial nerves III, IV, and VI. Semin Neurol. 2009;29:14-28. http://www.ncbi.nlm.nih.gov/pubmed/19214929?tool=bestpractice.com
The abducens nucleus contains motor neurons for the lateral rectus and interneurons traveling through the medial longitudinal fasciculus to the contralateral third nerve nucleus (to allow simultaneous movement of the contralateral medial rectus muscle). The nerve fascicles leave the nucleus and travel within the pontine tegmentum to leave the brainstem in the horizontal sulcus between the pons and medulla. The nerve enters the subarachnoid space and courses vertically along the clivus over the petrous apex of the temporal bone, where it is tethered in the Dorello canal. It then enters the cavernous sinus lateral to the internal carotid artery and finally enters the orbit through the superior orbital fissure.[9]Brazis PW. Isolated palsies of cranial nerves III, IV, and VI. Semin Neurol. 2009;29:14-28. http://www.ncbi.nlm.nih.gov/pubmed/19214929?tool=bestpractice.com
Function and disorders
The third, fourth, and sixth cranial nerves are responsible for eye movements.
The third cranial nerve controls most extraocular muscles, including the superior, inferior, and medial recti, and the inferior oblique muscles. In addition, it innervates the levator palpebrae superioris, which elevates the eyelid, and carries parasympathetic innervation to the pupil. Patients often present with paralysis of adduction, elevation, and depression, and when the pupil is involved a large unreactive pupil is noted. This presentation can suggest serious neurologic disorders, namely subarachnoid hemorrhage, cerebral aneurysms, uncal herniation, or meningitis. Prompt recognition and evaluation is needed.
The fourth cranial nerve innervates the superior oblique muscle, which controls depression, intorsion, and adduction of the eye. It is the most common cause of vertical diplopia. The frequency of fourth nerve palsy is difficult to accurately report, but in one large series it was more common than both oculomotor and abducens palsies.[11]Holmes JM, Mutyala S, Maus TL, et al. Pediatric third, fourth, and sixth nerve palsies: a population-based study. Am J Ophthalmol. 1999;127:388-392. http://www.ncbi.nlm.nih.gov/pubmed/10218690?tool=bestpractice.com [12]Richards BW, Jones FR Jr, Younge BR. Causes and prognosis in 4,278 cases of paralysis of the oculomotor, trochlear, and abducens cranial nerves. Am J Ophthalmol. 1992;113:489-496. http://www.ncbi.nlm.nih.gov/pubmed/1575221?tool=bestpractice.com The abducens nerve innervates the lateral rectus muscle and controls abduction. Patients typically present with horizontal double vision. It may be an isolated finding or part of a systemic disease.[9]Brazis PW. Isolated palsies of cranial nerves III, IV, and VI. Semin Neurol. 2009;29:14-28. http://www.ncbi.nlm.nih.gov/pubmed/19214929?tool=bestpractice.com
Nerve testing
Simple bedside testing of eye movements can be performed to elicit a third, fourth, or sixth nerve palsy. The patient is asked to keep his or her head still and follow the examiner's index finger with the eyes. The examiner slowly moves his or her finger up and down and from side to side at eye level and observes eye movements.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com The patient should report back any diplopia. Diplopia is maximal in the direction of action of the paralyzed muscle. Of the duplicated images perceived by the patient, the image seen in the periphery is perceived from the paretic (abnormal) eye and should disappear upon occlusion of the eye.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com
Trigeminal (V)
Anatomy
The trigeminal nerve has 3 main branches: ophthalmic (V1), maxillary (V2), and mandibular (V3).[13]Nemzek WR. The trigeminal nerve. Top Magn Reson Imaging. 1996;8:132-154. http://www.ncbi.nlm.nih.gov/pubmed/8840469?tool=bestpractice.com V1 enters the cranial cavity through the superior orbital fissure, V2 through the foramen rotundum, and V3 through the foramen ovale. V1 and V2 traverse the cavernous sinus. The first-order cell bodies carrying modalities of pain, temperature, pressure, and light touch in all 3 branches are located in the trigeminal (gasserian) ganglion in the Meckel cave (near the petrous apex of the temporal bone). Proprioceptive fibers have their first-order cell bodies in the mesencephalic nucleus of the brainstem. From the trigeminal ganglion, the nerve fibers enter the pons and synapse in multiple trigeminal nuclei. From there, second-order neurons carry afferent information to the ventral posteromedial nucleus of the thalamus. Finally, third-order neurons relay to the primary sensory cortex. Efferent motor fibers originate in the motor nucleus of the trigeminal nerve in the midpons and travel with V3 through the foramen ovale to supply the muscles of mastication (masseter, temporalis, mylohyoid, medial and lateral pterygoid, and anterior belly of the digastric), as well as the tensor tympani and tensor veli palatini. The trigeminal nerve and its branches also mediate the afferent limbs of the corneal blink and lacrimal reflexes, and both afferent and efferent limbs of the jaw-jerk reflex.
Function and disorders
The trigeminal nerve is the largest cranial nerve.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com It carries sensation from the face and mucosal surfaces, cornea, and supratentorial dura, as well as providing motor innervations to the muscles of mastication. The differential diagnosis for trigeminal neuropathy is very broad. Intra-axial pathology, particularly of the pons, can result in trigeminal dysfunction, but only rarely does this result in a mononeuropathy. Extra-axial lesions are more likely to affect the trigeminal nerve or its branches alone. Symptoms of trigeminal neuropathy depend on the location and etiology of the lesion and may include loss of sensation in the distribution of one or more trigeminal nerve branches, neuropathic pain, or weakness of the muscles of mastication.[14]Hughes MA, Frederickson AM, Branstetter BF, et al. MRI of the trigeminal nerve in patients with trigeminal neuralgia secondary to vascular compression. AJR Am J Roentgenol. 2016 Mar;206(3):595-600. https://www.ajronline.org/doi/10.2214/AJR.14.14156 http://www.ncbi.nlm.nih.gov/pubmed/26901017?tool=bestpractice.com
Nerve testing
Facial sensation can be tested by asking the patient to close his or her eyes and report where a stimulus is felt. Light touch with a cotton wool stick, pinprick with the end of a sterile needle, and warm and cold stimuli can be tested on each side of the face.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com Contraction of the masseter and temporal muscles can be examined by visual inspection, and palpation of the masseter muscles can be examined when the patient is chewing.
The jaw jerk can be tested as follows: with the patient's mouth slightly open, the mandible is tapped just below the lips in a downward direction. The masseter will move the mandible upward. Normally this reflex is weak, but it may be pronounced with upper motor neuron lesions.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com
The strength of the pterygoid muscles may be tested by asking the patient to open the jaw against resistance.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com
The corneal reflex can be tested with cotton wool (afferent-trigeminal, efferent-facial) and elicits an ipsilateral and contralateral blink response in normal individuals.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com
Electrodiagnostic testing: the afferent component of the trigeminal nerve (V1) may be evaluated via the blink reflex. Needle electromyography of trigeminal nerve-innervated muscles, such as masseter and temporalis, tests for the motor efferent component of the trigeminal nerve (V3). Less common tests of the trigeminal nerve include masseter inhibitory reflex and jaw jerk (the latter is similar to the aforementioned physical exam).[8]Rubin DI. Clinical neurophysiology. 5th ed. Oxford: Oxford University Press; 2021.[15]Muzyka IM, Estephan B. Electrophysiology of cranial nerve testing: trigeminal and facial nerves. J Clin Neurophysiol. 2018 Jan;35(1):16-24. http://www.ncbi.nlm.nih.gov/pubmed/29298209?tool=bestpractice.com [16]Kennelly KD. Electrodiagnostic approach to cranial neuropathies. Neurol Clin. 2012 May;30(2):661-84. http://www.ncbi.nlm.nih.gov/pubmed/22361379?tool=bestpractice.com [17]Perotto AO. Anatomical guide for the electromyographer: the limbs and trunk. 5th ed. Springfield, IL: Charles C Thomas Publisher; 2011.
Facial (VII)
Anatomy
The facial nerve is composed of both motor and sensory roots (nervus intermedius) and has a long intracranial course with 3 bends and multiple branches. The motor root has neuronal cell bodies in the facial nucleus of the lateral caudal pons. Fibers from the nucleus course posteriorly and form a sharp loop around the sixth nerve nucleus, forming the facial colliculus. The seventh cranial nerve then exits the brainstem at the pontomedullary junction, traverses the cerebellopontine cistern, and enters the facial canal through the meatus of the internal auditory canal. The nervus intermedius carries general somatic afferent and special visceral efferent fibers, and is separate from the motor root only between the brainstem and the facial canal. The geniculate ganglion, containing the cell bodies of general somatic afferent and special visceral efferent neurons, is located in the temporal bone within the facial canal.
The first branch of the seventh cranial nerve is the greater superficial petrosal nerve, which travels to the sphenopalatine and pterygopalatine ganglion, and carries parasympathetic fibers to innervate the lacrimal gland of the eye. The second branch innervates the stapedius muscle. The chorda tympani (third branch) carries taste sensation from the anterior two-thirds of the tongue, as well as parasympathetic innervation to the sublingual and submandibular glands (through the submandibular ganglion). The facial nerve exits the cranium through the stylomastoid foramen and enters the parotid gland, where it splits into 5 terminal branches (temporal, zygomatic, buccal, mandibular, and cervical), which innervate the muscles of facial expression, and digastric and stylohyoid muscles.
Function and disorders
Facial nerve mononeuropathy is the most common cranial nerve mononeuropathy. It can affect people of all ages. There are many etiologies, and the most important initial step is to rule out central causes of facial weakness, including ischemic stroke and pontine neoplasms.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com The most common peripheral facial palsy is Bell palsy.[18]Libreros-Jiménez HM, Manzo J, Rojas-Durán F, et al. On the cranial nerves. NeuroSci. 2024 Mar;5(1):8-38. https://pmc.ncbi.nlm.nih.gov/articles/PMC11523702 http://www.ncbi.nlm.nih.gov/pubmed/39483811?tool=bestpractice.com
Nerve testing
Muscles of facial expression can be tested to determine facial nerve function. Patients are asked to close their eyes tightly and resist opening, raise their eyebrows against resistance, show their teeth, and purse their lips. With a unilateral upper motor neuron lesion, such as a stroke, only the lower half of the face on the contralateral side is affected, due to bilateral innervation of the upper facial muscles.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com With a lower motor neuron lesion, such as a Bell palsy, there is ipsilateral upper and lower face weakness.
Electrodiagnostic testing: a combination of facial nerve motor conduction studies, blink reflex testing, and needle electromyography of facial muscles in at least 4 of the 5 terminal branches (temporal, zygomatic, buccal, and marginal mandibular) may be used to determine the extent and severity of facial nerve dysfunction.[15]Muzyka IM, Estephan B. Electrophysiology of cranial nerve testing: trigeminal and facial nerves. J Clin Neurophysiol. 2018 Jan;35(1):16-24. http://www.ncbi.nlm.nih.gov/pubmed/29298209?tool=bestpractice.com [16]Kennelly KD. Electrodiagnostic approach to cranial neuropathies. Neurol Clin. 2012 May;30(2):661-84. http://www.ncbi.nlm.nih.gov/pubmed/22361379?tool=bestpractice.com [17]Perotto AO. Anatomical guide for the electromyographer: the limbs and trunk. 5th ed. Springfield, IL: Charles C Thomas Publisher; 2011.
Neuromuscular ultrasonography: the facial nerve can be visualized with high-frequency ultrasonography underneath the ear lobe, and as it enters the parotid gland. This is technically challenging (the facial nerve can be isoechoic [the same echotexture] as the parotid gland), but can be used to assess for compression, focal nerve changes, and nerve continuity.[19]Tawfik EA, Walker FO, Cartwright MS. Neuromuscular ultrasound of cranial nerves. J Clin Neurol. 2015 Apr;11(2):109-21. https://thejcn.com/DOIx.php?id=10.3988/jcn.2015.11.2.109 http://www.ncbi.nlm.nih.gov/pubmed/25851889?tool=bestpractice.com
Vestibulocochlear (VIII)
Anatomy
Cell bodies of the vestibular division reside in the vestibular (Scarpa) ganglion in the internal acoustic meatus. Their dendrites project to the hair cells of the vestibular sensory organs (hair cells in the ampullae of the 3 semicircular canals, and hair cells in the maculae of the utricle and saccule) and axons project to the lateral, medial, superior, and inferior vestibular nuclei in the caudal pons. The cochlear division of the eighth cranial nerve has cell bodies in the spiral (auditory) ganglion with dendrites projecting to the hair cells of the auditory sensory organ (the organ of Corti within the cochlea). Axons of the cochlear division exit the internal acoustic meatus and course with the vestibular portion to enter the brainstem at the junction of the pons and medulla (cerebellopontine angle), and synapse in the ventral and dorsal cochlear nuclei of the rostral medulla.
Function and disorders
The vestibulocochlear nerve is a purely special sensory afferent nerve consisting of vestibular and cochlear divisions. Their axons run together through the internal acoustic meatus (which also transmits the facial nerve) and the brainstem. Symptoms of dysfunction include hearing loss, tinnitus, and vertigo.
Nerve testing
Simple bedside hearing tests such as whispering a word or number in one ear with the other covered and having the patient repeat the word can be used to assess the degree of hearing impairment.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com
Rinne test: a tuning fork is placed on the mastoid bone (bone conduction) until the sound can no longer be heard. The tuning fork is then placed next to the external ear (air conduction). Usually air conduction is better than bone conduction, so the sound can still be heard; this is a positive Rinne test. If bone conduction is better than air conduction, this is a negative Rinne test and indicates conductive hearing loss in that ear.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com
Weber test: the tuning fork is placed on the forehead. The patient is asked in which ear the sound is louder. If the patient hears the sound equally in each ear or cannot localize, this is normal and is termed a midline Weber. The Weber lateralizes toward a conductive hearing loss and away from a sensorineural hearing loss. For example, if the patient hears the sound louder in the right ear, then this indicates either a right conductive or a left sensorineural hearing loss.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com
Electrodiagnostic testing: brainstem auditory evoked potentials (BAEPs) may be performed to identify and localize auditory pathway lesions. This potential is recorded from surface electrodes on the patient’s head, while sound is repeatedly played over a headphone or earpiece in either ear.[8]Rubin DI. Clinical neurophysiology. 5th ed. Oxford: Oxford University Press; 2021. Use of BAEPs has declined due to widespread availability of neuroimaging.
Glossopharyngeal (IX)
Anatomy
The glossopharyngeal nerve exits the rostral medulla at the pontomedullary junction, crosses the cerebellopontine cistern, and exits the cranial cavity through the jugular foramen. It contains general somatic afferent, general visceral efferent, special visceral afferent, and parasympathetic fibers that innervate the tongue and pharynx.[20]Erman AB, Kejner AE, Hogikyan ND, et al. Disorders of cranial nerves IX and X. Semin Neurol. 2009;29:85-92. http://www.ncbi.nlm.nih.gov/pubmed/19214937?tool=bestpractice.com Visceral and taste fibers within the nerve end in the nucleus solitarius of the medulla, which also receives afferent fibers from the carotid body and carotid sinus. Preganglionic parasympathetic fibers travel through the tympanic nerve to the lesser petrosal nerve and synapse in the otic ganglion before supplying the parotid gland.
Function and disorders
It is predominantly a sensory nerve but also contains some motor and parasympathetic fibers. Isolated glossopharyngeal neuropathy is rare, as lesions often involve other cranial nerves in close proximity (VIII, X, XI, and XII).[20]Erman AB, Kejner AE, Hogikyan ND, et al. Disorders of cranial nerves IX and X. Semin Neurol. 2009;29:85-92. http://www.ncbi.nlm.nih.gov/pubmed/19214937?tool=bestpractice.com Additionally, isolated palsy of the glossopharyngeal nerve can often be asymptomatic, due to redundant innervation of target structures by other cranial nerves. The nerve innervates the tongue and pharynx, including pain, temperature, and tactile sensation from the posterior third of the tongue, the tonsils, medial tympanic membrane, and Eustachian tube. It also innervates the stylopharyngeus muscle, involved in swallowing, mediates taste from the posterior third of the tongue, and sends parasympathetic innervation to the parotid gland.[21]Snell RS. Clinical neuroanatomy. Philadelphia, PA: Lippincott Williams & Wilkins; 2001.[22]Remley KB, Harnsberger HR, Smoker WR, et al. CT and MRI in the evaluation of glossopharyngeal, vagal, and spinal accessory neuropathy. Semin Ultrasound CT MR. 1987;8:284-300.
Nerve testing
The gag reflex is absent if a nerve palsy is present, as the afferent impulse is carried by the glossopharyngeal nerve.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com
Vagus (X)
Anatomy
The vagus nerve exits the brainstem just below the glossopharyngeal nerve, at the pontomedullary junction, traverses the cerebellopontine angle, and exits the cranium through the jugular foramen. The first main branch of the vagus nerve is the pharyngeal branch, which runs in the carotid sheath between the internal and external carotid arteries. It innervates the levator veli palatini, salpingopharyngeus, and palatopharyngeus muscles, and the uvula.[23]Hendelman W. Atlas of functional neuroanatomy. 3rd ed. Boca Raton, FL: CRC Press; 2015. The superior laryngeal nerve descends lateral to the pharynx; its external branch innervates the cricothyroid muscle. The recurrent laryngeal nerve is the third motor branch of the vagus and supplies all intrinsic muscles of the larynx except the cricothyroid. The right recurrent laryngeal nerve loops around the right subclavian artery, while the left loops under the aortic arch before ascending in the tracheoesophageal groove to the larynx.[24]Castillo M, Mukherji SK. Magnetic resonance imaging of cranial nerves IX, X, XI, and XII. Top Magn Reson Imaging. 1996;8:180-186. http://www.ncbi.nlm.nih.gov/pubmed/8840472?tool=bestpractice.com
Function and disorders
The vagus nerve contains both visceral efferent and afferent fibers and has 3 main motor branches.[21]Snell RS. Clinical neuroanatomy. Philadelphia, PA: Lippincott Williams & Wilkins; 2001. It innervates all striated muscles of the larynx and pharynx, except the stylopharyngeus muscle (innervated by IX) and the tensor veli palatini muscle (mandibular branch of V). Sensory input from the larynx, pharynx, external auditory canal, lateral tympanic membrane, and posterior fossa meningeal layers are mediated by the vagus.[25]Parent A. Carpenter's human neuroanatomy. 9th ed. Baltimore, MD: Lippincott Williams & Wilkins; 1996. Visceral afferent information is also conveyed by the vagus nerve from the thoracic and abdominal viscera, and it delivers parasympathetic fibers to these regions as well, in addition to the larynx and pharynx.[18]Libreros-Jiménez HM, Manzo J, Rojas-Durán F, et al. On the cranial nerves. NeuroSci. 2024 Mar;5(1):8-38. https://pmc.ncbi.nlm.nih.gov/articles/PMC11523702 http://www.ncbi.nlm.nih.gov/pubmed/39483811?tool=bestpractice.com
Nerve testing
Tenth nerve palsy can result in hoarseness, dysphagia, and dyspnea, as well as palatal droop and deviation of the uvula to the contralateral side.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com [18]Libreros-Jiménez HM, Manzo J, Rojas-Durán F, et al. On the cranial nerves. NeuroSci. 2024 Mar;5(1):8-38. https://pmc.ncbi.nlm.nih.gov/articles/PMC11523702 http://www.ncbi.nlm.nih.gov/pubmed/39483811?tool=bestpractice.com Lesions distal to pharyngeal branches, or a lesion of the recurrent laryngeal nerve itself, present with isolated hoarseness. The gag reflex is absent, as the efferent limb is formed by the vagus nerve.[18]Libreros-Jiménez HM, Manzo J, Rojas-Durán F, et al. On the cranial nerves. NeuroSci. 2024 Mar;5(1):8-38. https://pmc.ncbi.nlm.nih.gov/articles/PMC11523702 http://www.ncbi.nlm.nih.gov/pubmed/39483811?tool=bestpractice.com
Electrodiagnostic testing: electromyographers experienced in laryngeal needle placement can perform laryngeal electromyography to assess for laryngeal nerve disorder.[17]Perotto AO. Anatomical guide for the electromyographer: the limbs and trunk. 5th ed. Springfield, IL: Charles C Thomas Publisher; 2011.[26]Smith LJ, Munin MC. Utility of laryngeal electromyography for establishing prognosis and individualized treatment after laryngeal neuropathies. Muscle Nerve. 2024 Jul 30. https://onlinelibrary.wiley.com/doi/10.1002/mus.28207 http://www.ncbi.nlm.nih.gov/pubmed/39080992?tool=bestpractice.com
Neuromuscular ultrasonography: the vagus nerve can be visualized with high-frequency ultrasonography in the carotid sheath in the neck at the level of the thyroid, typically in between the carotid artery and internal jugular vein. Sonographic assessment of the nerve at this site can be used to see focal nerve changes, assessing for a demyelinating polyradiculopathy.
Spinal accessory (XI)
Anatomy
The spinal accessory nerve originates in the rostral spinal cord at C1 to C5 levels through a series of rootlets that emerge between the dorsal and ventral roots. It ascends through the foramen magnum and exits the skull through the jugular foramen. The fibers of the spinal accessory nerve emerge through the posterior border of the sternocleidomastoid (SCM) muscle and supply both the SCM and the trapezius muscles. As it crosses the posterior triangle of the neck it is closely related to the superficial cervical lymph nodes.[27]Massey EW. Spinal accessory nerve lesions. Semin Neurol. 2009;29:82-84. http://www.ncbi.nlm.nih.gov/pubmed/19214936?tool=bestpractice.com
Historically, the spinal accessory nerve and the “cranial root of the accessory nerve” were identified as constituents of a single “accessory nerve.” The cranial root originates from the nucleus ambiguous and exits from the lateral aspect of the medulla, briefly joining the spinal accessory nerve before it separates to join the vagus nerve, and innervating the muscles of the soft palate, larynx, and pharynx. Because of its functional similarity, the cranial root is commonly grouped as part of the vagus nerve.[18]Libreros-Jiménez HM, Manzo J, Rojas-Durán F, et al. On the cranial nerves. NeuroSci. 2024 Mar;5(1):8-38. https://pmc.ncbi.nlm.nih.gov/articles/PMC11523702 http://www.ncbi.nlm.nih.gov/pubmed/39483811?tool=bestpractice.com [28]Johal J, Iwanaga J, Tubbs K, et al. The accessory nerve: a comprehensive review of its anatomy, development, variations, landmarks and clinical considerations. Anat Rec (Hoboken). 2019 Apr;302(4):620-9. https://anatomypubs.onlinelibrary.wiley.com/doi/10.1002/ar.23823 http://www.ncbi.nlm.nih.gov/pubmed/29659160?tool=bestpractice.com
Function and disorders
The spinal accessory nerve is purely a motor nerve and supplies the SCM and trapezius muscles.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com It is not commonly injured, but due to its long, superficial extracranial course, it is susceptible to iatrogenic injury.[28]Johal J, Iwanaga J, Tubbs K, et al. The accessory nerve: a comprehensive review of its anatomy, development, variations, landmarks and clinical considerations. Anat Rec (Hoboken). 2019 Apr;302(4):620-9. https://anatomypubs.onlinelibrary.wiley.com/doi/10.1002/ar.23823 http://www.ncbi.nlm.nih.gov/pubmed/29659160?tool=bestpractice.com Injury to this nerve can result in lateral scapular winging and unilateral inability to shrug the shoulder.
Nerve testing
The spinal accessory nerve can be assessed by testing the strength of trapezius and SCM muscles. Trapezius weakness results in a drooping shoulder at rest and mild lateral scapular winging with attempted shoulder elevation and arm abduction >90°. When the patient shrugs his or her shoulders against resistance, unilateral weakness may be detected.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com SCM weakness results in difficulty when turning the head in the opposite direction to the injury. To test, the patient is asked to turn his or her head to the side against resistance.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com Proximal lesions of the nerve produce weakness of both the SCM and trapezius muscles.
Electrodiagnostic testing: motor nerve conduction studies of the spinal accessory nerve as well as needle electromyography of the trapezius and/or SCM may be performed to assess for spinal accessory neuropathy.[17]Perotto AO. Anatomical guide for the electromyographer: the limbs and trunk. 5th ed. Springfield, IL: Charles C Thomas Publisher; 2011.
Neuromuscular ultrasonography: the spinal accessory nerve can be visualized with high-frequency ultrasonography in the posterior triangle of the neck. This is a technically challenging, but can be used to assess for compression, focal nerve changes, and nerve continuity. Muscle ultrasonography of the SCM and/or three portions of the trapezius muscle (upper, middle, and lower) in the setting of a spinal accessory neuropathy can reveal atrophy, muscular changes, and lack of movement with dynamic assessment.[19]Tawfik EA, Walker FO, Cartwright MS. Neuromuscular ultrasound of cranial nerves. J Clin Neurol. 2015 Apr;11(2):109-21. https://thejcn.com/DOIx.php?id=10.3988/jcn.2015.11.2.109 http://www.ncbi.nlm.nih.gov/pubmed/25851889?tool=bestpractice.com [29]Krzesniak-Swinarska M, Caress JB, Cartwright MS. Neuromuscular ultrasound for evaluation of scapular winging. Muscle Nerve. 2017 Jul;56(1):7-14. http://www.ncbi.nlm.nih.gov/pubmed/28006862?tool=bestpractice.com
Hypoglossal (XII)
Anatomy
The hypoglossal nucleus is located in the dorsal aspect of the caudal medulla, just below the floor of the fourth ventricle. General somatic efferent fibers from the nuclei course ventrally to exit the brainstem as two bundles of rootlets at the ventrolateral sulcus of the medial medulla. These rootlets exit the cranium through the hypoglossal canal just rostral to the foramen magnum and unite on the extracranial side. The hypoglossal nerve descends lateral to the internal carotid artery and vagus nerve, and then courses anteriorly to supply the ipsilateral intrinsic and extrinsic (genioglossus, styloglossus, and hyoglossus) muscles of the tongue. Additionally, efferent motor fibers from C1 course along the hypoglossal nerve for a short distance before leaving to innervate the geniohyoid and thyrohyoid muscles, and form the superior root of the ansa cervicalis. Meningeal branches from C1 and C2 innervating the dura mater of the posterior fossa are also carried by the hypoglossal nerve.[30]Standring S. Gray's anatomy: the anatomical basis of clinical practice. 42nd ed. Amsterdam: Elsevier; 2020. Finally, the hypoglossal nerve receives sympathetic fibers from the superior cervical ganglion and communicates with the lingual branch of the mandibular nerve, which mediates tactile sensation from the anterior two-thirds of the tongue.[18]Libreros-Jiménez HM, Manzo J, Rojas-Durán F, et al. On the cranial nerves. NeuroSci. 2024 Mar;5(1):8-38. https://pmc.ncbi.nlm.nih.gov/articles/PMC11523702 http://www.ncbi.nlm.nih.gov/pubmed/39483811?tool=bestpractice.com [31]Lalwani AK. Current diagnosis and treatment in otolaryngology: head and neck surgery. 4th ed. New York, NY: McGraw-Hill; 2019.
Function and disorders
The 12th nerve is purely motor in function. It moves and alters the shape of the tongue by providing ipsilateral motor innervation to the intrinsic and extrinsic tongue muscles.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com Lesions of the medulla (nuclear lesions) can cause 12th nerve dysfunction but are usually associated with other neurologic symptoms and cranial nerve deficits.
Nerve testing
A 12th nerve mononeuropathy can be due to nuclear (medullary) or infranuclear lesions. Close exam of the tongue at rest and in motion can help establish this diagnosis. Symptoms of a 12th nerve palsy typically include unilateral or bilateral tongue weakness with deviation toward the affected side on tongue protrusion, tongue atrophy (with scalloping or accentuation of the midline groove), fasciculation of the tongue at rest, tongue flaccidity, or the inability to rapidly move the tongue side to side or vertically.[4]Damodaran O, Rizk E, Rodriguez J, et al. Cranial nerve assessment: a concise guide to clinical examination. Clin Anat. 2014 Jan;27(1):25-30. https://www.doi.org/10.1002/ca.22336 http://www.ncbi.nlm.nih.gov/pubmed/24307604?tool=bestpractice.com
Electrodiagnostic testing: needle electromyography of the intrinsic tongue muscles, such as the genioglossus muscle, may be performed to assess for hypoglossal neuropathy or motor neurone disease.[17]Perotto AO. Anatomical guide for the electromyographer: the limbs and trunk. 5th ed. Springfield, IL: Charles C Thomas Publisher; 2011.[32]Preston DC, Shapiro BE. Electromyography and neuromuscular disorders: clinical-electrophysiologic-ultrasound correlations. 4th ed. Philadelphia, PA: Elsevier; 2021.
Neuromuscular ultrasonography: muscle ultrasonography of the intrinsic tongue muscles is a more sensitive tool than needle electromyography to reveal tongue fasciculations in motor neurone disease. Focal changes in the echotexture of the hypoglossal-innervated muscles can be visualized; however, the hypoglossal nerve is not readily seen.[33]Grimm A, Prell T, Décard BF, et al. Muscle ultrasonography as an additional diagnostic tool for the diagnosis of amyotrophic lateral sclerosis. Clin Neurophysiol. 2015 Apr;126(4):820-7. http://www.ncbi.nlm.nih.gov/pubmed/25204706?tool=bestpractice.com
Differentials
Common
- Giant cell arteritis (II, III, IV, VI)
- Non-arteritic anterior ischemic optic neuropathy (II)
- Multiple sclerosis (II)
- Viral infection (II)
- Subarachnoid hemorrhage (III, IV, VI)
- Meningitis (III, IV, VI)
- Vascular malformations (V)
- Herpes zoster (V)
- Multiple sclerosis (V)
- Bell palsy (VII)
- Ramsay Hunt syndrome (VII)
- Ischemic stroke (VII)
- Vestibular neuritis (VIII)
- Neural presbycusis (VIII)
- Drugs (VIII)
- Iatrogenic (X)
- Apical lung tumor (IX, X)
- Iatrogenic (XI)
- Ischemic stroke (XII)
Uncommon
- Trauma (I)
- Neurodegenerative disorders (I)
- Congenital (I)
- CNS tumors (I)
- Optic canal trauma (II)
- CNS tumors (II)
- Idiopathic intracranial hypertension (II)
- Autoimmune disease: (e.g., systemic lupus erythematosus (SLE), Sjogren, granulomatosis with polyangiitis, Behcet disease [II])
- Leber hereditary optic neuropathy (II)
- Optical toxins or nutritional deficiency (II)
- Neuromyelitis optica (II)
- Uncal herniation (III, IV, VI)
- Migraine (III, IV, VI)
- Trauma (III, IV, VI)
- Cerebral aneurysms (III, IV, VI)
- Cavernous-carotid fistula (III, IV, VI)
- Cavernous sinus thrombus (III, IV, VI)
- CNS tumors (III, IV, VI)
- Drugs (III, IV, VI)
- Idiopathic intracranial hypertension (III, IV, VI)
- Congenital (III, IV, VI)
- Post-lumbar puncture (VI)
- Meningitis (V)
- CNS tumors (V)
- Autoimmune disorders (V)
- Skull-base osteomyelitis (V)
- Trauma (V)
- Dental abscess (V)
- Spinal cord lesion (V)
- Iatrogenic (V)
- Mandibular tumors (V)
- Congenital (V)
- Tolosa-Hunt syndrome (V)
- Wallenberg syndrome (V)
- Neurosarcoidosis (VII)
- CNS tumors (VII)
- Trauma (VII)
- Meningitis (VII)
- Iatrogenic (VII)
- Middle ear or mastoid infection (VII)
- Parotid tumor (VII)
- HIV associated (VII)
- Lyme disease (VII)
- CNS tumors (VIII)
- CNS tumors (IX, X)
- Parapharyngeal tumor (IX, X)
- Meningitis (IX, X)
- Skull-base osteomyelitis (IX, X)
- Trauma (IX, X)
- Parapharyngeal space infection (IX, X)
- Eagle syndrome (IX)
- Cardiovocal syndrome (X)
- Trauma (XI)
- CNS tumors (XI)
- CNS tumors (XII)
- Motor neurone disease/Progressive bulbar palsy (XII)
- Chiari I and II malformations (XII)
- Extracranial (tongue or neck) or skull-base tumors (XII)
- Meningitis (XII)
- Skull-base osteomyelitis (XII)
- Parapharyngeal space infection (XII)
- Trauma (XII)
- Dural arteriovenous fistula (XII)
- Internal carotid artery aneurysm or dissection (XII)
- Iatrogenic (XII)
Contributors
Authors
James B Meiling, DO, RMSK
Assistant Professor
Department of Physical Medicine and Rehabilitation
Mayo Clinic
Rochester
MN
Disclosures
JBM declares that he has no competing interests.
Tatsuya Oishi, MD
Assistant Professor
Department of Neurology
Mayo Clinic
Rochester
MN
Disclosures
TO declares that he has no competing interests.
Acknowledgements
Dr Meiling and Dr Oishi would like to gratefully acknowledge Dr Ann Johnston, Dr Emma Tallantyre, Dr Zachary L. Hickman, Dr Brad E. Zacharia, Dr Christopher J. Winfree, and Dr Adrian J. Wills, the previous contributors to this topic.
Disclosures
AJ has received speaker fees from UCB and Eisai. ET has received honoraria for consulting work from Novartis, Merck, Biogen, and Roche, and received travel grants to attend or speak at educational meetings from Merck, Roche, Takeda, and Novartis. ZLH, BEZ, CJW, and AJW declare that they have no competing interests.
Peer reviewers
Benjamin D. Gallagher, MD, FACP
Assistant Professor of Medicine
Yale School of Medicine
New Haven
CT
Disclosures
BDG declares that he has no competing interests.
Sabrina Ravaglia, MD, PhD
Staff Physician
Department of Neurological Sciences
Institute of Neurology C. Mondino
Pavia
Italy
Disclosures
SR declares that she has no competing interests.
Peer reviewer acknowledgements
BMJ Best Practice topics are updated on a rolling basis in line with developments in evidence and guidance. The peer reviewers listed here have reviewed the content at least once during the history of the topic.
Disclosures
Peer reviewer affiliations and disclosures pertain to the time of the review.
References
Key articles
Libreros-Jiménez HM, Manzo J, Rojas-Durán F, et al. On the cranial nerves. NeuroSci. 2024 Mar;5(1):8-38.Full text Abstract
Erman AB, Kejner AE, Hogikyan ND, et al. Disorders of cranial nerves IX and X. Semin Neurol. 2009;29:85-92. Abstract
Gronseth G, Cruccu G, Alksne J, et al. Practice parameter: the diagnostic evaluation and treatment of trigeminal neuralgia (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology and the European Federation of Neurological Societies. Neurology. 2008 Oct 7;71(15):1183-90.Full text Abstract
American College of Radiology. ACR appropriateness criteria: cranial neuropathy. 2022 [internet publication].Full text
Reference articles
A full list of sources referenced in this topic is available to users with access to all of BMJ Best Practice.
Patient information
Migraine
More Patient informationLog in or subscribe to access all of BMJ Best Practice
Use of this content is subject to our disclaimer