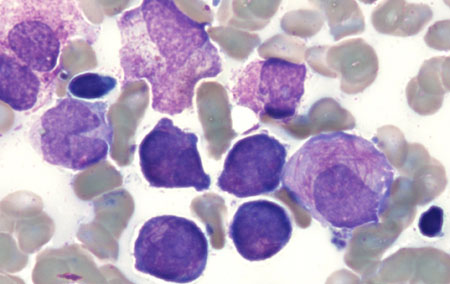
Summary
Definition
History and exam
Key diagnostic factors
- pallor
- ecchymoses or petechiae
Other diagnostic factors
- fatigue
- dizziness
- palpitations
- dyspnea
- fever and infections
- lymphadenopathy
- hepatosplenomegaly
- mucosal bleeding
- testicular mass
- skin mass (e.g., myeloid sarcoma)
- skin infiltration
- gingival enlargement
- bone pain
- gastrointestinal symptoms (e.g., severe abdominal pain)
- neurologic symptoms (e.g., headache, confusion)
Risk factors
- age over 65 years
- previous treatment with chemotherapy
- previous hematologic disorders
- inherited genetic disorders
- constitutional chromosomal abnormalities
- ionizing radiation exposure
- benzene exposure
- environmental exposures
- male sex
Diagnostic tests
1st tests to order
- CBC with differential
- peripheral blood smear
- coagulation panel
- serum electrolytes
- serum uric acid
- serum lactate dehydrogenase (LDH)
- renal function
- liver function tests
- bone marrow evaluation
- genetic testing
Tests to consider
- genetic testing for heritable hematologic malignancy predisposition syndrome
- human leukocyte antigen (HLA) typing
- CNS imaging and lumbar puncture
- fluorodeoxyglucose-PET/CT scan
- CXR
- chest CT
- echocardiogram
- multigated acquisition scan
- human leukocyte antigen (HLA) typing
Treatment algorithm
newly diagnosed AML: suitable for intensive chemotherapy
newly diagnosed AML: not suitable for intensive chemotherapy
newly diagnosed non-high-risk acute promyelocytic leukemia (APL)
newly diagnosed high-risk acute promyelocytic leukemia (APL)
complete remission: AML
complete remission: acute promyelocytic leukemia (APL)
relapsed or refractory AML
relapsed or refractory acute promyelocytic leukemia (APL)
Contributors
Authors
Vijaya Raj Bhatt, MBBS, MS
Associate Professor
Section Leader, Malignant Hematology
University of Nebraska Medical Center Division of Hematology-Oncology
Nebraska
NE
Disclosures
VRB has participated in a Safety Monitoring Committee for Protagonist Therapeutics. He serves as an Associate Editor for the Elsevier Journal, Current Problems in Cancer. He has received consulting fees from Imugene, Sanofi, and Taiho; research funding (institutional) from Abbvie, Pfizer, Incyte, Jazz, NMDP, MEI Pharma, Sanofi, and Actinium Pharmaceuticals; and drug support (institutional) from Chimerix for a trial.
Prajwal Dhakal, MBBS
Clinical Assistant Professor of Internal Medicine-Hematology, Oncology, and Blood and Marrow Transplantation
University of Iowa
Iowa City
IA
Disclosures
PD declares that he has no competing interests.
Acknowledgements
Dr Vijaya Raj Bhatt and Dr Prajwal Dhakal would like to gratefully acknowledge Dr Kavita Raj and Dr Priyanka Mehta, previous contributors to this topic.
Disclosures
KR declares that she has no competing interests. PM is an author of a reference cited in this topic.
Peer reviewers
Naveen Premnath, MD
Assistant Professor of Medicine
Division of Hematology, Oncology, and Transplantation
University of Minnesota
Minnesota
MN
Disclosures
NP declares that he has no competing interests.
Rebecca Connor, MD
Chief Fellow
Section of Hematology and Oncology
Department of Internal Medicine
Wake Forest University Baptist Medical Center
Winston-Salem
NC
Disclosures
RC declares that she has no competing interests.
Roger M. Lyons, MD, FACP
Clinical Professor of Medicine
University of Texas Health Science Center
San Antonio
Cancer Care Network of South Texas
San Antonio
TX
Disclosures
RML declares that he has no competing interests.
Shankaranarayana Paneesha, MD, MRCP, FRCPath
Consultant Haematologist
Department of Haematology and Stem Cell Transplantation
Heartlands Hospital
Birmingham
UK
Disclosures
SP declares that he has no competing interests.
David Marks, MD, MRCP, MRCPath
Professor of Haematology & Stem Cell Transplantation
Department of Molecular and Cellular Medicine
University of Bristol
UK
Disclosures
DM declares that he has no competing interests.
Peer reviewer acknowledgements
BMJ Best Practice topics are updated on a rolling basis in line with developments in evidence and guidance. The peer reviewers listed here have reviewed the content at least once during the history of the topic.
Disclosures
Peer reviewer affiliations and disclosures pertain to the time of the review.
References
Key articles
Döhner H, Wei AH, Appelbaum FR, et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood. 2022 Sep 22;140(12):1345-77.Full text Abstract
National Cancer Comprehensive Network. NCCN guidelines in oncology: acute myeloid leukemia [internet publication].Full text
Heuser M, Ofran Y, Boissel N, et al. Acute myeloid leukaemia in adult patients: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2020 Jun;31(6):697-712.Full text Abstract
Sanz MA, Fenaux P, Tallman MS, et al. Management of acute promyelocytic leukemia: updated recommendations from an expert panel of the European LeukemiaNet. Blood. 2019 Apr 11;133(15):1630-43.Full text Abstract
Sekeres MA, Guyatt G, Abel G, et al. American Society of Hematology 2020 guidelines for treating newly diagnosed acute myeloid leukemia in older adults. Blood Adv. 2020 Aug 11;4(15):3528-49.Full text Abstract
Reference articles
A full list of sources referenced in this topic is available to users with access to all of BMJ Best Practice.
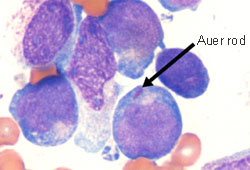
Differentials
- Acute lymphoblastic leukemia (ALL)
- Biphenotypic leukemia
- Myeloid/lymphoid neoplasms with eosinophilia and tyrosine kinase fusion genes (MLNE)
More DifferentialsGuidelines
- NCCN guidelines in oncology: acute myeloid leukemia
- NCCN practice guidelines in oncology: hematopoietic cell transplantation
More GuidelinesLog in or subscribe to access all of BMJ Best Practice
Use of this content is subject to our disclaimer