Summary
Definition
History and exam
Key diagnostic factors
- blistering skin lesions
Other diagnostic factors
- skin hyperpigmentation
- hypertrichosis
- scarring alopecia
- red urine
Risk factors
- male, middle-aged, white people
- alcohol use
- smoking
- estrogen therapy
- hepatitis C
- HIV
- hereditary hemochromatosis gene (HFE) mutation
- uroporphyrinogen decarboxylase (UROD) mutations
- exposure to halogenated polycyclic aromatic hydrocarbons
- reduced levels of antioxidants
- end-stage renal disease
- diabetes mellitus
Diagnostic tests
1st tests to order
- plasma total porphyrins
- plasma fluorescence emission
- urinary total porphyrins
- erythrocyte total porphyrins
Tests to consider
- fractionation of plasma porphyrins by high-performance liquid chromatography (HPLC)
- fractionation of urinary porphyrins by HPLC
- erythrocyte uroporphyrinogen decarboxylase (UROD) activity
- fecal porphyrins
- DNA studies
- Liver function tests
- serum ferritin
- liver biopsy
- skin biopsy
- serum HIV enzyme-linked immunosorbent assay
- serum hepatitis C surface antibodies
- creatinine
- BUN
- hematocrit
- hemoglobin
Treatment algorithm
no phlebotomy contraindications
phlebotomy contraindicated or poorly tolerated
relapse after remission
Contributors
Authors
Gagan Sood, MD
Associate Professor
Department of Medicine and Surgery
Baylor College of Medicine
Houston
TX
Disclosures
GS is an author of a number of references cited in this topic.
Karl E. Anderson, MD
Professor
Departments of Preventive Medicine and Community Health and Internal Medicine
University of Texas Medical Branch
Galveston
TX
Disclosures
KEA has received grants from the National Institutes of Health, the US Food and Drug Administration, and Alnylam Pharmaceuticals; he is an author of a number of references cited in this topic. KEA has received consulting fees, advisory board fees, and grants to the university from Alnylam Pharmaceuticals; consulting fees, advisory board fees, and grants from Recordati Rare Diseases; and consulting fees and grants from Mitsubishi Tanabe Pharma America.
Peer reviewers
Robert S. Dawe, MBChB, MRCP(UK), MD(Glasgow)
Consultant Dermatologist
Honorary Clinical Senior Lecturer
Department of Dermatology
Ninewells Hospital & Medical School
Dundee
UK
Disclosures
RSD declares that he has no competing interests.
Jeffrey P. Callen, MD
Professor of Medicine (Dermatology)
University of Louisville
Louisville
KY
Disclosures
JPC declares that he has no competing interests.
Montgomery Bissell, MD
Professor and Chief
Gastroenterology
University of California
San Francisco
CA
Disclosures
MB declares that he has no competing interests.
Peer reviewer acknowledgements
BMJ Best Practice topics are updated on a rolling basis in line with developments in evidence and guidance. The peer reviewers listed here have reviewed the content at least once during the history of the topic.
Disclosures
Peer reviewer affiliations and disclosures pertain to the time of the review.
References
Key articles
Phillips JD, Bergonia HA, Reilly CA, et al. A porphomethene inhibitor of uroporphyrinogen decarboxylase causes porphyria cutanea tarda. Proc Natl Acad Sci USA. 2007 Jan;104:5079-84.Full text Abstract
Jalil S, Grady JJ, Lee C, et al. Associations among behavior-related susceptibility factors in porphyria cutanea tarda. Clin Gastroenterol Hepatol. 2010 Mar;8(3):297-302;e1.Full text Abstract
Singal AK. Porphyria cutanea tarda: recent update. Mol Genet Metab. 2019 Nov;128(3):271-81. Abstract
Handler NS, Handler MZ, Stephany MP, et al. Porphyria cutanea tarda: an intriguing genetic disease and marker. Int J Dermatol. 2017 Jun;56(6):e106-17. Abstract
Reference articles
A full list of sources referenced in this topic is available to users with access to all of BMJ Best Practice.
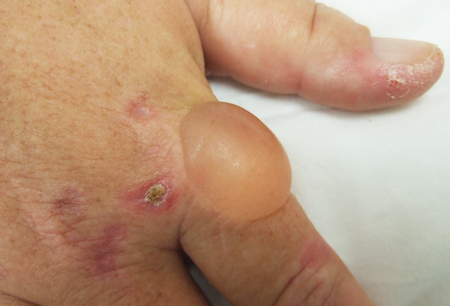
Differentials
- Variegate porphyria (VP)
- Hereditary coproporphyria (HCP)
- Congenital erythropoietic porphyria (CEP)
More DifferentialsLog in or subscribe to access all of BMJ Best Practice
Use of this content is subject to our disclaimer