Summary
Definition
History and exam
Key diagnostic factors
- fever or history of fever
Other diagnostic factors
- headache
- weakness
- myalgia
- arthralgia
- anorexia
- diarrhea
- seizures
- nausea and vomiting
- abdominal pain
- pallor
- hepatosplenomegaly
- jaundice
- altered level of consciousness
- hypotension
- bleeding
- anuria/oliguria
- tachypnea
Risk factors
- travel to endemic area
- inadequate or absent chemoprophylaxis
- insecticide-treated bed net not used in endemic area
- low host immunity (severe disease)
- pregnancy (severe disease)
- age <5 years (severe disease)
- immunocompromise (severe disease)
- older age (severe disease)
- malnutrition (severe disease)
- iron administration (children)
Diagnostic tests
1st tests to order
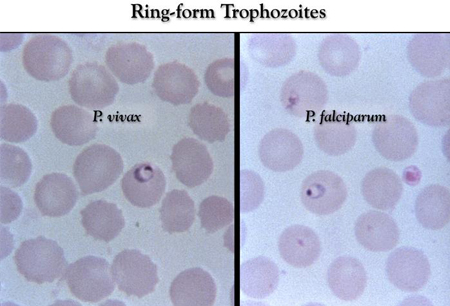
- Giemsa-stained thick and thin blood smears
- rapid diagnostic tests (RDTs)
- CBC
- clotting profile
- serum electrolytes, BUN and creatinine
- serum LFTs
- serum blood glucose
- urinalysis
- arterial blood gas
Tests to consider
- polymerase chain reaction (PCR) blood for malaria
- chest x-ray
- blood culture
- urine culture
- sputum culture
- lumbar puncture
- HIV test
- PCR nasopharyngeal swabs for influenza or COVID-19
- CT head
Emerging tests
- loop-mediated isothermal amplification
Treatment algorithm
severe disease (or unable to take oral treatment initially): all Plasmodium species
Plasmodium falciparum (or unknown species): uncomplicated disease
Plasmodium ovale: uncomplicated disease
Plasmodium vivax: uncomplicated disease
Plasmodium malariae or Plasmodium knowlesi: uncomplicated disease
Plasmodium falciparum: recurrent infection
Contributors
Authors
Elizabeth Ashley, MB BS, FRCP, FRCPath
Director
Institution Lao-Oxford-Mahosot Hospital - Wellcome Trust Research Unit
Vientiane
Laos
Honorary Consultant in Infectious Diseases and Microbiology
Oxford University Hospitals NHS Foundation Trust
Professor of Tropical Medicine
University of Oxford
Oxford
UK
Disclosures
EA declares that she has no competing interests. EA is an author of a number of references cited in this topic.
Arjun Chandna, BA MRCP AFHEA
Clinical Research Fellow
Centre for Tropical Medicine and Global Health
University of Oxford
Oxford
Specialty Registrar in Infectious Diseases and Medical Microbiology
University College London Hospitals NHS Trust
London
UK
Disclosures
AC declares that he has no competing interests.
Acknowledgements
Dr Elizabeth Ashley and Dr Arjun Chandna would like to gratefully acknowledge Professor Ron Behrens, Mariyam Mirfenderesky, Dr Signe Maj Sorensen, Dr Joanna Allen, Dr Simon Warren, and Dr Behzad Nadjm, previous contributors to this topic.
Disclosures
RB acted as a paid expert to the courts on malaria prophylaxis. RB received fees on the Travel Health advisory board for Emergent BioSolutions. RB prepared education material for the Royal College of Physicians and Surgeons of Glasgow. RB is an author of a number of references cited in this topic. MM, SMS, JA, and SW declare that they have no competing interests. BN is an author of a reference cited in this topic.
Peer reviewers
Blaise Genton, MD
Professor
Head of the Travel Clinic
Consultant of Tropical and Travel Medicine
University Hospital
Project Leader
Swiss Tropical and Public Health Institute
Basel
Switzerland
Disclosures
BG has received a research grant from Novartis Pharma to assess the impact of the introduction of artemether-lumefantrine (Novartis) as first-line treatment for uncomplicated malaria on mortality of children under 5 years old in 2 districts in Tanzania and travel grants from Novartis Pharma to present the results of the study above. BG is an author of a reference cited in this topic.
David Sullivan, MD
Associate Professor
Malaria Research Institute and Department of Molecular Microbiology and Immunology
Johns Hopkins Bloomberg School of Public Health
Baltimore
MD
Disclosures
DS has received royalties from antigen provision for a diagnostic test to Inverness. DS with Johns Hopkins University has patents on diagnostic tests that do not require blood.
Walther H. Wernsdorfer, MD
Professor
Institute of Specific Prophylaxis and Tropical Medicine
Medical University of Vienna
Vienna
Austria
Disclosures
WHW declares that he has no competing interests.
Peer reviewer acknowledgements
BMJ Best Practice topics are updated on a rolling basis in line with developments in evidence and guidance. The peer reviewers listed here have reviewed the content at least once during the history of the topic.
Disclosures
Peer reviewer affiliations and disclosures pertain to the time of the review.
References
Key articles
World Health Organization. WHO guidelines for malaria. Aug 2025 [internet publication].Full text
Centers for Disease Control and Prevention. Clinical guidance: malaria diagnosis & treatment in the U.S. Jun 2024 [internet publication].Full text
Reference articles
A full list of sources referenced in this topic is available to users with access to all of BMJ Best Practice.
Differentials
- Dengue fever
- Zika virus infection
- Chikungunya virus infection
More DifferentialsGuidelines
- Malaria control in emergencies: field manual
- WHO guidelines for malaria
More GuidelinesPatient information
Malaria prevention
More Patient informationVideos
Diagnostic lumbar puncture in adults: animated demonstration
More videosLog in or subscribe to access all of BMJ Best Practice
Use of this content is subject to our disclaimer