Summary
Definition
History and exam
Key diagnostic factors
- history of intensive chemotherapy regimens
- history of radiation therapy to the oral cavity
- history of chemoradiation
- erythema and/or ulceration of oral mucosa
- oral pain
Other diagnostic factors
- intraoral bleeding
- dietary impairment and/or weight loss
- diarrhea
- nausea and/or vomiting
- abdominal pain
- fever
Risk factors
- intensive chemotherapy regimens
- radiation therapy to the oral cavity
- chemoradiation
- genetic polymorphisms in drug metabolic enzymes
- use of targeted therapies and immunotherapies
Diagnostic tests
1st tests to order
- clinical diagnosis
Tests to consider
- CBC with differential
- blood cultures
- superficial smear of lesion for microscopy
- fungal culture
- viral culture or polymerase chain reaction (PCR)
Treatment algorithm
undergoing hematopoietic stem cell transplant: preventive measures
receiving bolus fluorouracil: preventive measures
receiving radiation therapy to oral cavity: preventive measures
established oral mucositis
Contributors
Authors
Ourania Nicolatou-Galitis, DDS, MSc, DrDent
Dentist
Specialist in Oral Medicine and Oral Oncology
Hospital Dentistry
National & Kapodistrian University of Athens
Athens
Greece
Disclosures
ONG has received fees from Angelini pharma to author a review article on oral mucositis and is the cofounder and scientific responsible of mycancer, a startup private company which aims to provide a comprehensive medical platform for cancer patients. ONG is an author of several references cited in this topic.
Nikolaos Tsoukalas, MD, MSc, PhD
Medical Oncologist
Associate Director
Department of Oncology
401 General Military Hospital
Athens
Greece
Disclosures
NT declares that he has no competing interests.
Acknowledgements
Professor Ourania Nicolatou-Galitis and Dr Nikolaos Tsoukalas would like to gratefully acknowledge Dr Eleni Arvanitou, who contributed to the updating of this topic, and Dr Rajesh V. Lalla, a previous contributor.
Disclosures
EA declares that she has no competing interests. RVL serves as a consultant for Galera Therapeutics, Ingalfarma, Mundipharma and Sucampo Pharma, and has received research funding from Onxeo, Sucampo Pharma, Galera Therapeutics, and Novartis.
Peer reviewers
Michael Brennan, DDS, MHS
Associate Chairman and Oral Medicine Residency Director
Department of Oral Medicine
Carolinas Medical Center
Charlotte
NC
Disclosures
MB is an author of a reference cited in this topic. MB is part of the Mucositis Study Group of MASCC/ISOO, and participated in the review of mucositis.
Maria Michelagnoli, MB ChB, MD, FRCPCH
Consultant Pediatric and Adolescent Oncologist
University College London Hospitals
London
UK
Disclosures
MM declares that she has no competing interests.
Peer reviewer acknowledgements
BMJ Best Practice topics are updated on a rolling basis in line with developments in evidence and guidance. The peer reviewers listed here have reviewed the content at least once during the history of the topic.
Disclosures
Peer reviewer affiliations and disclosures pertain to the time of the review.
References
Key articles
Elad S, Cheng KKF, Lalla RV, et al. MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer. 2020 Oct 1;126(19):4423-31.Full text Abstract
Peterson DE, Boers-Doets CB, Bensadoun RJ, et al; ESMO Guidelines Committee. Management of oral and gastrointestinal mucosal injury: ESMO clinical practice guidelines for diagnosis, treatment, and follow-up. Ann Oncol. 2015 Sep;26(suppl 5):v139-51.Full text Abstract
Reference articles
A full list of sources referenced in this topic is available to users with access to all of BMJ Best Practice.
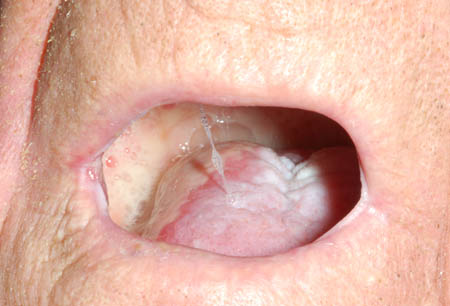
Differentials
- Oral candidiasis
- Herpes simplex virus infection
- Graft-versus-host disease
More DifferentialsGuidelines
- MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy
- Management of oral and gastrointestinal mucosal injury: ESMO clinical practice guidelines
More GuidelinesLog in or subscribe to access all of BMJ Best Practice
Use of this content is subject to our disclaimer