Summary
Definition
History and exam
Key diagnostic factors
- presentation in infancy
Other diagnostic factors
- tachypnea/shortness of breath (SOB)
- faltering growth
- exercise intolerance
- widened pulse pressure
- machine-like continuous murmur/Gibson murmur in children born at full-term
- apnea
- low diastolic blood pressure (BP)
- irritability
- diaphoresis
- increased respiratory symptoms with upper respiratory infection
- murmur heard only during systole
- hyperdynamic precordium
- systolic thrill
- third heart sound heard at apex
- mid-diastolic rumble heard at apex
- bounding peripheral pulses
- pulmonary rales
Risk factors
- prematurity
- maternal rubella
- female sex
- respiratory distress syndrome (RDS)
- high altitude
- family history
- black race
Diagnostic tests
1st tests to order
- CXR
- ECG
- echocardiogram
Tests to consider
- cardiac catheterization and angiography
Treatment algorithm
premature infants: hemodynamically stable
premature infants: hemodynamically significant
premature infants: contraindication to pharmacotherapy or persistent hemodynamic significance unresponsive to initial pharmacotherapy
term infants and children: small-to-moderate-sized ducts
term infants and children: large ducts and/or symptomatic infants too small for device closure
adults
Contributors
Authors
Kenneth M. Coca, MD
Pediatric Cardiology Fellow
Johns Hopkins All Children’s Hospital
Saint Petersburg
FL
Disclosures
KMC declares that he has no competing interests.
Joyce T. Johnson, MD, MS
Director, Fetal Cardiology
Johns Hopkins All Children’s Hospital
Saint Petersburg
FL
Disclosures
JTJ declares that she has no competing interests.
Acknowledgements
Dr Kenneth M. Coca and Dr Joyce T. Johnson would like to gratefully acknowledge Dr Anji T. Yetman and Dr Nelangi M. Pinto, previous contributors to this topic.
Disclosures
ATY declares that she has no competing interests.
Peer reviewers
Michael Cheung, BSc (Hons), MB ChB, MRCP, FRACP
Acting Head
Department of Cardiology
Royal Children's Hospital
Melbourne
Australia
Disclosures
MC declares that he has no competing interests.
Rajat Bhatt, MD
Assistant Professor
Internal Medicine
Texas Tech University Health Sciences Center (TTUHSC)
Lubbock
TX
Disclosures
RB declares that he has no competing interests.
Henry M. Sondheimer, MD
Professor of Pediatrics
Clinical Director
Pediatric Cardiology
The Children's Hospital
Denver
CO
Disclosures
HMS declares that he has no competing interests.
Peer reviewer acknowledgements
BMJ Best Practice topics are updated on a rolling basis in line with developments in evidence and guidance. The peer reviewers listed here have reviewed the content at least once during the history of the topic.
Disclosures
Peer reviewer affiliations and disclosures pertain to the time of the review.
References
Key articles
Baumgartner H, De Backer J, Babu-Narayan SV, et al. 2020 ESC guidelines for the management of adult congenital heart disease. Eur Heart J. 2021 Feb 11;42(6):563-645.Full text
Ambalavanan N, Aucott SW, Salavitabar A, et al. Patent ductus arteriosus in preterm infants. Pediatrics. 2025 May 1;155(5):e2025071425.Full text Abstract
Wilson WR, Gewitz M, Lockhart PB, et al. Prevention of viridans group streptococcal infective endocarditis: a scientific statement from the American Heart Association. Circulation. 2021 May 18;143(20):e963-78.Full text Abstract
Stout KK, Daniels CJ, Aboulhosn JA, et al. 2018 AHA/ACC guideline for the management of adults with congenital heart disease: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2019 Apr 2;73(12):1494-563.Full text Abstract
John AS, Jackson JL, Moons P, et al. Advances in managing transition to adulthood for adolescents with congenital heart disease: a practical approach to transition program design: a scientific statement from the American Heart Association. J Am Heart Assoc. 2022 Apr 5;11(7):e025278.Full text Abstract
Reference articles
A full list of sources referenced in this topic is available to users with access to all of BMJ Best Practice.
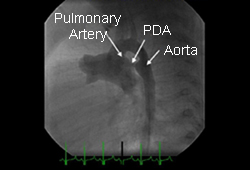
Differentials
- Venous hum
- Coronary artery fistula
- Left-sided shunts (ventricular septal defect, atrioventricular septal defect)
More DifferentialsGuidelines
- Patent ductus arteriosus in preterm infants
- Pulmonary hypertension in congenital heart disease: a scientific statement
More GuidelinesLog in or subscribe to access all of BMJ Best Practice
Use of this content is subject to our disclaimer