Summary
Definition
History and exam
Key diagnostic factors
- milk, egg, nut, fish, shellfish, wheat, or soy ingestion
- reproducible symptoms
- flushing, urticaria, or angioedema of the skin
- sneezing, rhinorrhea, or nasal congestion
- dyspnea, tachypnea, wheezing, coughing, or cyanosis
- hoarseness, stridor, or sense of choking
- nausea and vomiting
- abdominal cramping or bloating
- diarrhea
- conjunctival injection or lacrimation
- periorbital edema
- abrupt onset of symptoms
- reaction caused by small amount of food
- presence of other allergic disease
- laryngeal edema
Other diagnostic factors
- tachycardia or bradycardia
- reaction exacerbated by exercise or exertion
- alcohol or drug ingestion before reaction
- cardiac arrhythmia
- hypotension
Risk factors
- family history of food allergy
- atopic dermatitis
- newborn
- perinatal peanut oil exposure
Diagnostic investigations
1st investigations to order
- in vitro IgE-specific immunoassay
- skin prick testing
Tests to avoid
- IgG testing
Investigations to consider
- food challenges
- component-resolved diagnostics
Treatment algorithm
anaphylactic reaction
cutaneous symptoms
bronchospasm
rhinoconjunctivitis
following stabilization
Contributors
Authors
A. Wesley Burks, MD

Curnen Distinguished Professor and Chair
Department of Pediatrics
University of North Carolina
Chapel Hill
NC
Disclosures
AWB receives grant support to his institution from the National Institutes of Health and the Burroughs Wellcome Fund; royalties from UpToDate, Elsevier, and Walter Kluwer; consulting honorariums from Astella Pharma Global Development, Allergy Therapeutics (UK) Ltd, DBV Technologies, Kaléo, N-Fold, LLC, ALK-Abelló Inc, and UKKO Inc, as well as Aimmune Therapeutics, Consortia TX Inc, and Prota Therapeutics for his service on their respective scientific advisory boards. AWB owns stock in Allertein and Mastcell Pharmaceuticals. These interests do not directly relate to this topic but are being shared for full disclosure. AWB is an author of several references cited in this topic.
J. Andrew Bird, MD
Associate Professor
Department of Pediatrics
Division of Allergy and Immunology
University of Texas Southwestern Medical Center
Dallas
TX
Disclosures
JAB consults for AllerGenis, Allergy Therapeutics Ltd, Before Brands, DBV Technologies, Genentech, and Novartis. He receives grant funding to his institution from Aimmune, DBV Technologies, Genentech, HIH-NIAD, Novartic, Siolta, and Regeneron. JAB is the author of one reference cited in this topic.
Peer reviewers
Justin Skripak, MD
Assistant Professor of Pediatric Allergy and Immunology
Mount Sinai School of Medicine
New York
NY
Disclosures
JS declares that he has no competing interests.
Hugh A. Sampson, MD
Professor of Pediatrics
Mount Sinai School of Medicine
New York
NY
Disclosures
HAS holds a 4% interest in a biotech company, Allertein Pharmaceuticals LLC, which is developing an engineered recombinant protein vaccine for peanut allergy, and 45% interest in a virtual company, Herbal Springs LLC, that holds a patent application on a herbal product for treating asthma and another for treating food allergy. HAS is an author of several references cited in this topic.
Adam Fox, MA(Hons) Cantab., MSc, MBBS, DCH, FRCPCH, FHEA, Dip. Allergy
Consultant and Honorary Senior Lecturer in Paediatric Allergy
Evelina Children's Hospital
Guy's & St Thomas' Hospitals NHS Foundation Trust
London
UK
Disclosures
AF declares that he has no competing interests.
Peer reviewer acknowledgements
BMJ Best Practice topics are updated on a rolling basis in line with developments in evidence and guidance. The peer reviewers listed here have reviewed the content at least once during the history of the topic.
Disclosures
Peer reviewer affiliations and disclosures pertain to the time of the review.
References
Key articles
Chafen JJ, Newberry SJ, Riedl MA, et al. Diagnosing and managing common food allergies: a systematic review. JAMA. 2010 May 12;303(18):1848-56. Abstract
Sicherer SH, Sampson HA. Food allergy: recent advances in pathophysiology and treatment. Annu Rev Med. 2009 Feb;60:261-77. Abstract
Greer FR, Sicherer SH, Burks AW, et al. The effects of early nutritional interventions on the development of atopic disease in infants and children: the role of maternal dietary restriction, breastfeeding, hydrolyzed formulas, and timing of introduction of allergenic complementary foods. Pediatrics. 2019 Apr;143(4):e20190281.Full text Abstract
Muraro A, Halken S, Arshad SH, et al. EAACI food allergy and anaphylaxis guidelines: primary prevention of food allergy. Allergy. 2014 May;69(5):590-601.Full text Abstract
Sampson HA, Gerth van Wijk R, Bindslev-Jensen C, et al. Standardizing double-blind, placebo-controlled oral food challenges: American Academy of Allergy, Asthma & Immunology-European Academy of Allergy and Clinical Immunology PRACTALL consensus report. J Allergy Clin Immunol. 2012 Dec;130(6):1260-74.Full text Abstract
Heyman MB. American Academy of Pediatrics, Committee on Nutrition. Lactose intolerance in infants, children, and adolescents. Pediatrics. 2006 Sep;118(3):1279-86.Full text Abstract
Reference articles
A full list of sources referenced in this topic is available to users with access to all of BMJ Best Practice.
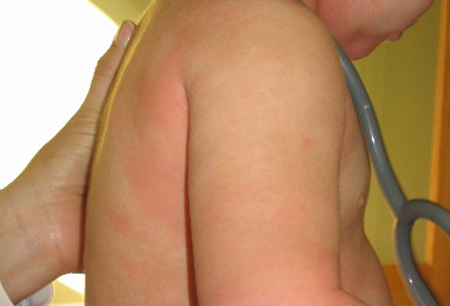
Differentials
- Atopic dermatitis
- Urticaria
- Auriculotemporal syndrome
More DifferentialsGuidelines
- Diagnosis and management of celiac disease
- Anaphylaxis - a 2023 practice parameter update
More GuidelinesPatient information
Food allergy: what is it?
Food allergy: what treatments are available?
More Patient informationLog in or subscribe to access all of BMJ Best Practice
Use of this content is subject to our disclaimer