Resumo
Definição
História e exame físico
Principais fatores diagnósticos
- rectal bleeding
- diarrhea
- blood in stool
Outros fatores diagnósticos
- abdominal pain
- arthritis and spondylitis
- malnutrition
- abdominal tenderness
- fever
- weight loss
- constipation
- skin rash
- uveitis and episcleritis
- pallor
Fatores de risco
- family history of inflammatory bowel disease
- human leukocyte antigen-B27
- infection
- use of nonsteroidal anti-inflammatory drugs (NSAIDs)
- not smoking or former smoker
Investigações diagnósticas
Primeiras investigações a serem solicitadas
- stool studies for infective pathogens
- fecal calprotectin (FC)
- CBC
- comprehensive metabolic panel (including LFTs)
- erythrocyte sedimentation rate (ESR)
- CRP
- plain abdominal radiograph
- flexible sigmoidoscopy
- colonoscopy
- biopsies
Tests to avoid
- serologic antibody testing
Investigações a serem consideradas
- CT scan
- intestinal ultrasound
Algoritmo de tratamento
acute severe ulcerative colitis (UC)
moderate-to-severe disease
mild disease
disease in remission
Colaboradores
Autores
Andrew Poullis, BSc, MBBS, MD, FRCP
Consultant Gastroenterologist
Department of Gastroenterology
St George’s Hospital
London
UK
Declarações
AP is an author of a reference cited in this topic.
Sailish Honap, MBChB (Hons), MRes, MRCP (UK)
Consultant Gastroenterologist
Department of Gastroenterology
St George’s Hospital
London
UK
Declarações
SH has received honoraria from Pfizer (speaker, consultant, advisory board member, and received travel grants), Janssen (speaker fees, travel grants), AbbVie (speaker fees, consultant, travel grants), Takeda (speaker fees), Ferring (travel grants), Pharmacosmos (travel grants), Galapagos (travel grants).
Kamal Patel,
Declarações
all disclosures
Aaron Bhakta, BMedSci (Hons), BMBS, MSc, MRCP
Clinical Fellow in Gastroenterology
Department of Gastroenterology
St George’s Hospital
London
UK
Declarações
AB declares that he has no competing interests.
Agradecimentos
Dr Hesam A. Nooredinvand, and Dr Andrew Poullis would like to gratefully acknowledge Dr Ben Shandro, Dr Anet Soubieres, Dr Rhys Hewett, Dr Willem J.S. de Villiers, and Dr Houssam Mardini, previous contributors to this topic.
Declarações
BS, AS, RH, WJSD, and HM declare that they have no competing interests.
Revisores
Stephen B Hanauer, MD
Professor of Medicine
Gastroenterology and Hepatology
Northwestern University
Evanston
IL
Declarações
SW declares that he has no competing interests.
Créditos aos pareceristas
Os tópicos do BMJ Best Practice são constantemente atualizados, seguindo os desenvolvimentos das evidências e das diretrizes. Os pareceristas aqui listados revisaram o conteúdo pelo menos uma vez durante a história do tópico.
Declarações
As afiliações e declarações dos pareceristas referem--se ao momento da revisão.
Referências
Principais artigos
World Gastroenterology Organisation. Global guidelines: inflammatory bowel disease. Aug 2015 [internet publication].Texto completo
Rubin DT, Ananthakrishnan AN, Siegel CA, et al. ACG clinical guideline update: ulcerative colitis in adults. Am J Gastroenterol. 2025 Jun 3;120(6):1187-224.Texto completo Resumo
Moran GW, Gordon M, Sinopolou V, et al. British Society of Gastroenterology guidelines on inflammatory bowel disease in adults: 2025. Gut. 2025 Jun 23;74(Suppl 2):s1-101.Texto completo Resumo
Singh S, Loftus EV Jr, Limketkai BN, et al. AGA living clinical practice guideline on pharmacological management of moderate-to-severe ulcerative colitis. Gastroenterology. 2024 Dec;167(7):1307-43.Texto completo Resumo
Harbord M, Eliakim R, Bettenworth D, et al. Third European evidence-based consensus on diagnosis and management of ulcerative colitis. Part 2: current management. J Crohns Colitis. 2017 Jul 1;11(7):769-84.Texto completo Resumo
National Institute for Health and Care Excellence (UK). Ulcerative colitis: management. May 2019 [internet publication].Texto completo
Artigos de referência
Uma lista completa das fontes referenciadas neste tópico está disponível para os usuários com acesso total ao BMJ Best Practice.
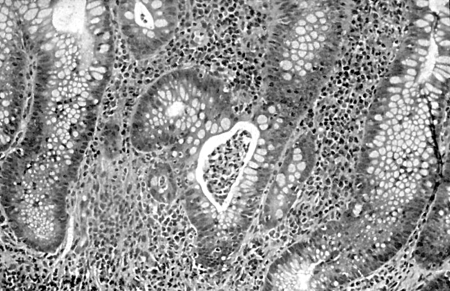
Diagnósticos diferenciais
- Crohn disease
- Indeterminate colitis
- Radiation colitis
Mais Diagnósticos diferenciaisDiretrizes
- Preventive care in inflammatory bowel disease
- Management of inflammatory bowel disease in adults
Mais DiretrizesFolhetos informativos para os pacientes
Ulcerative colitis: what is it?
Ulcerative colitis: what are the treatment options?
Mais Folhetos informativos para os pacientesVideos
Venepuncture and phlebotomy: animated demonstration
Mais vídeosConectar-se ou assinar para acessar todo o BMJ Best Practice
O uso deste conteúdo está sujeito ao nosso aviso legal