Summary
Definition
ანამნეზი და გასინჯვა
ძირითადი დიაგნოსტიკური ფაქტორები
- residence in/travel from endemic area
- fever
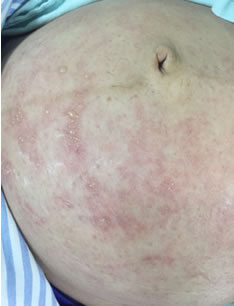
- maculopapular rash
- arthralgia
- conjunctivitis
- features of congenital Zika syndrome (infants)
- features of Guillain-Barre syndrome
სხვა დიაგნოსტიკური ფაქტორები
- other constitutional symptoms
- gastrointestinal symptoms
- retro-orbital pain
- edema of lower limbs
- transient hearing loss in adults
რისკფაქტორები
- residence in/travel from endemic area
- mosquito bites in endemic area
- unprotected sexual contact with infected individual
- blood transfusion from infected individual
- sperm donation from infected individual
- exposure to other infected body fluids
- exposure to infected human cells/tissues
დიაგნოსტიკური კვლევები
1-ად შესაკვეთი გამოკვლევები
- reverse transcriptase-polymerase chain reaction (RT-PCR)
- serology
- testing for other arboviruses
გასათვალისწინებელი კვლევები
- prenatal ultrasound
- amniocentesis
- head circumference measurement (newborn)
- newborn further evaluation
- CT/MRI head (newborn)
- tests for Guillain-Barre syndrome
მკურნალობის ალგორითმი
symptomatic: nonpregnant
pregnant: with possible mosquito-borne or sexual exposure
congenital Zika syndrome
Zika-associated Guillain-Barre syndrome
კონტრიბუტორები
ავტორები
Adriana Scavuzzi, MD, MSc, PhD
Coordinator Obstetrics/Gynecology
Microcephaly Working Group
Instituto de Medicina Integral Professor Fernando Figueira (IMIP)
Recife
Brazil
გაფრთხილება:
AS declares that she has no competing interests.
მადლიერება
Dr Adriana Scavuzzi would like to gratefully acknowledge Dr Geraldo Furtado, a previous contributor to this topic.
გაფრთხილება:
GF is an author of references cited in this topic.
რეცენზენტები
William A. Petri, Jr., MD, PhD
Chief
Division of Infectious Diseases and International Health
University of Virginia
Charlottesville
VA
გაფრთხილება:
WAP declares that he has no competing interests.
Dominic Dwyer, BSc (Med), MBBS, MD, FRACP, FRCPA
Director
Institute of Clinical Pathology and Medical Research
Pathology West
Westmead Hospital
Sydney
Australia
გაფრთხილება:
DD is a member of Advisory Boards for bioCSL and GSK in antivirals and vaccines.
Chad Achenbach, MD, MPH
Assistant Professor
Division of Infectious Diseases
Northwestern University Feinberg School of Medicine
Chicago
IL
გაფრთხილება:
CA declares that he has no competing interests.
Henry Wu, MD, DTM&H
Assistant Professor of Medicine
TravelWell Center
Division of Infectious Diseases
Atlanta
GA
გაფრთხილება:
HW declares that he has no competing interests.
რეცენზენტების განცხადებები
BMJ Best Practice-ის თემების განახლება სხვადასხვა პერიოდულობით ხდება მტკიცებულებებისა და რეკომენდაციების განვითარების შესაბამისად. ქვემოთ ჩამოთვლილმა რეცენზენტებმა თემის არსებობის მანძილზე კონტენტს ერთხელ მაინც გადახედეს.
გაფრთხილება
რეცენზენტების აფილიაციები და გაფრთხილებები მოცემულია გადახედვის მომენტისთვის.
წყაროები
ძირითადი სტატიები
Adebanjo T, Godfred-Cato S, Viens L, et al. Update: interim guidance for the diagnosis, evaluation, and management of infants with possible congenital Zika virus infection - United States, October 2017. MMWR Morb Mortal Wkly Rep. 2017 Oct 20;66(41):1089-99.სრული ტექსტი აბსტრაქტი
Pan American Health Organization; World Health Organization. Provisional remarks on Zika virus infection in pregnant women: document for health care professionals. Jan 2016 [internet publication].სრული ტექსტი
World Health Organization. Zika virus fact sheet. Dec 2022 [internet publication].სრული ტექსტი
Centers for Disease Control and Prevention. Zika virus: clinical testing and diagnosis for Zika virus disease. Feb 2025 [internet publication].სრული ტექსტი
გამოყენებული სტატიები
ამ თემაში მოხსენიებული წყაროების სრული სია ხელმისაწვდომია მომხმარებლებისთვის, რომლებსაც აქვთ წვდომა BMJ Best Practice-ის ყველა ნაწილზე.
Differentials
- Dengue fever
- Chikungunya virus
- West Nile virus
More DifferentialsGuidelines
- Clinical testing and diagnosis for Zika virus disease
- CDC Yellow Book: health information for international travel - Zika
More GuidelinesPatient information
Zika virus
More Patient informationLog in or subscribe to access all of BMJ Best Practice
Use of this content is subject to our disclaimer